THE IMPACT OF HEMATOPOIETIC STEM CELL TRANSPLANT AND PURE RED CELL APLASIA ON CEREBRAL HEMODYNAMICS IN AN ADULT WITH SICKLE CELL ANEMIA AND COMORBID MOYAMOYA SYNDROME
Melanie Leguizamon1, Wesley Richerson1, Adetola Kassim2, Manus J. Donahue1,3, Lori. C. Jordan4
1 Department of Neurology, Vanderbilt University Medical Center, Nashville, TN, USA
2 Department of Medicine, Division of Hematology-Oncology, Vanderbilt University Medical Center, Nashville, TN, USA
3 Department of Psychiatry and Behavioral Sciences, Vanderbilt University Medical Center, Nashville, TN, USA
4 Department of Pediatrics, Division of Pediatric Neurology, Vanderbilt University Medical Center, Nashville, TN, USA
Lori Jordan, MD, PhD
2200 Children’s Way DOT 11212
Vanderbilt University Medical Center
Nashville, TN 37232
Phone: 615-936-5536
Email: lori.jordan@vumc.org
Abstract
Background: An increasing number of patients with sickle cell anemia (SCA) receive curative hematopoietic-stem-cell-transplant (HSCT), which has been shown to normalize elevated cerebral blood flow (CBF) levels secondary to chronic hemolytic anemia. However, less information is available on how such reductions in CBF and improvements in hemoglobin level manifest in the setting of comorbid moyamoya syndrome (MMS).
Objective: We present a single case of a young adult female with history of SCA and secondary MMS with bilateral ischemic strokes, bilateral revascularization surgeries for MMS in adolescence, and a complex course following HSCT in her 20s. Our goal is to highlight the normalization of cerebral hemodynamics in response to transplant in a patient where such changes could be detrimental and secondarily to demonstrate the utility of advanced magnetic resonance imaging methods to monitor this response.
Methods: Non-contrast MRI of the brain was performed pre-transplant and approximately one-year, two-years, and three-years post-transplant. Cortical CBF and oxygen extraction fraction were assessed via arterial spin labeling and TRUST, respectively.
Results: From pre- to post-transplant and following treatment for pure red cell aplasia after ABO incompatible HSCT, the patient’s hematocrit improved from 30% to 41%, cortical CBF normalized from a moderately elevated cortical CBF of 66.2 ml/100g/min to 46.3 ml/100g/min, and oxygen extraction fraction (OEF), assessed by pseudocontinuous arterial spin labeling (pCASL), was maintained within an approximately normal range. No new cerebral infarcts developed during this time.
Conclusions: This complex post-transplant trajectory provides evidence of the potential for HSCT to normalize CBF in the setting of SCA and comorbid MMS without the development of new cerebral infarcts, and for CBF-weighted imaging methods having sensitivity to monitor the cerebral hemodynamic sequalae of anemic changes post-transplant.
Introduction
Sickle cell anemia (SCA), the most common and severe form of sickle cell disease (SCD), is caused by the inheritance of abnormal beta-globin alleles carrying the sickle mutation (homozygous HbSS phenotype) and associated chronic hemolytic anemia.1 In SCA, low blood oxygen content leads to compensatory cerebral hyperemia to maintain adequate oxygen delivery to tissue. When insufficient, this may lead to reduced cerebral oxygen delivery and associated infarction. Approximately 12% of SCD patients also develop moyamoya vasculopathy,2 characterized by intracranial steno-occlusion of primarily the anterior circulation, resulting in the development of lenticulostriate and leptomeningeal collateral vessels.3 Thus, patients with both moyamoya syndrome (MMS) and SCA are at a unique risk for stroke given their tenuous cerebral blood flow (CBF) due to severe intracranial stenosis and hemolytic anemia,4 which reduces the oxygen content of blood. Curative hematopoietic-stem-cell-transplant (HSCT) is now offered for SCA patients,5 increasing total hemoglobin while reducing hemoglobin S levels and largely normalizing compensatory hyperemia.6,7 However, less is known regarding whether significant perfusion pressure changes in response to curative HSCT alter collateral pathways and increase infarct risk in the setting of comorbid MMS. We report a case of surveillance anatomical and hemodynamic imaging in a patient with SCA and MMS before and after curative HSCT. This case supports the utility of neuroimaging in assessing CBF and OEF longitudinally and highlights the need for further investigation into the potential beneficial effects of HSCT in patients with comorbid presentation.
Case Presentation
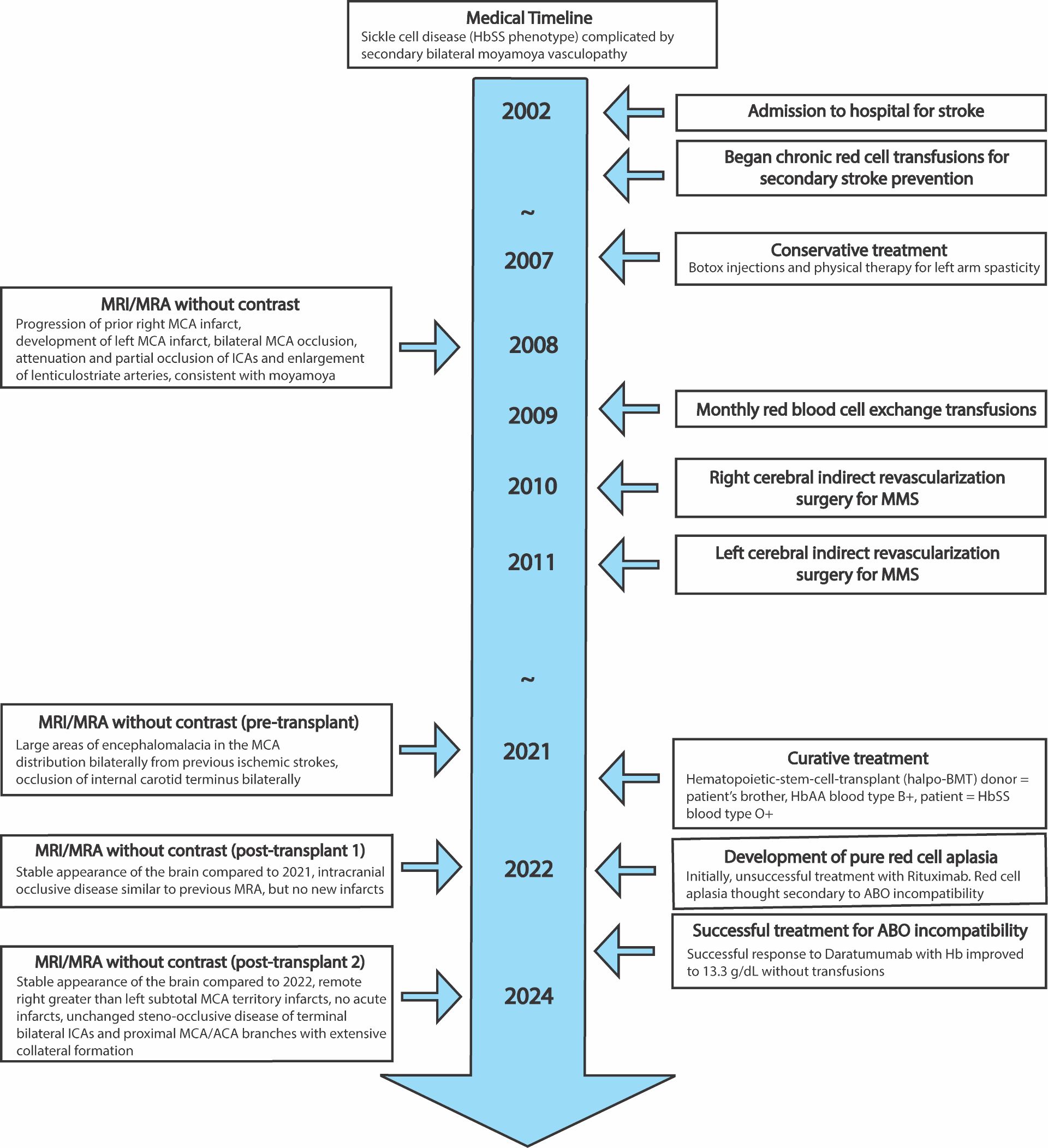
A female with SCA (HbSS) presented at six years-of-age with acute and chronic bilateral ischemic strokes (right>left frontoparietal lobes) in the setting of bacterial meningitis. Despite chronic red cell exchange therapy for secondary stroke prevention, she developed new cerebral infarctions, which has previously been documented in patients with sickle cell disease and vasculopathy.8 At age 13 years, she developed bilateral moyamoya vasculopathy with occlusions of the internal carotid artery (ICA) terminus bilaterally, as well as M1 and A1 partial occlusion and lenticulostriate artery enlargement. She subsequently underwent right and left indirect surgical revascularization with encephaloduroarteriosynangiosis at age 14 and 15 years, respectively, and demonstrated clinical stabilization. She continued to receive monthly exchange transfusions for many years (Figure 1).
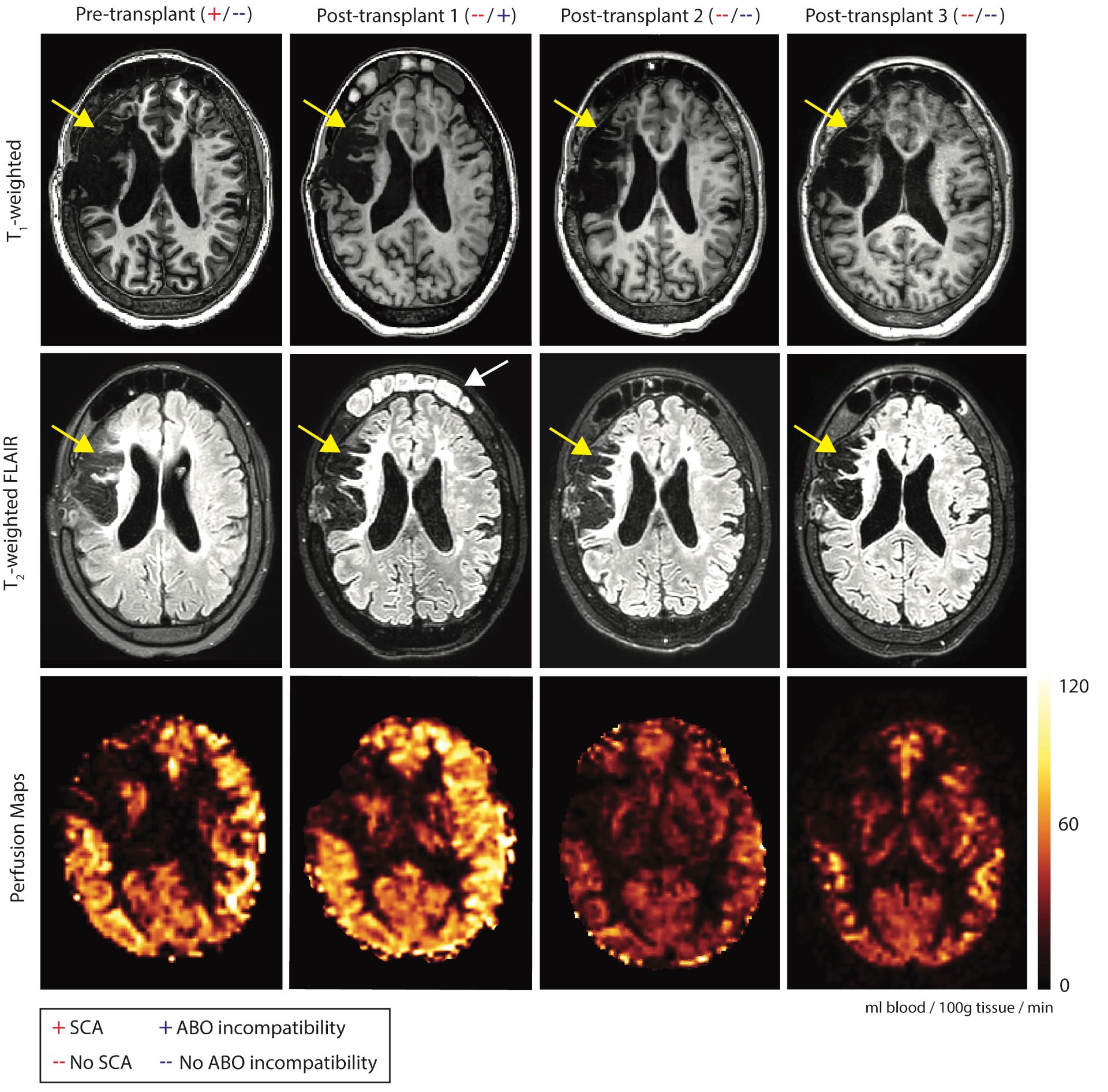
Figure 1
In her 20s, HSCT was recommended as curative treatment and was approved on an institutional transplant protocol due to stroke and MMS progression risk, and transfusion iron overload despite chelation. A non-myeloablative haploidentical bone marrow transplant (haplo-BMT) was prescribed, as the patient had no matched sibling and poor unrelated donor options. Her brother was identified as a haplo-match and served as the donor, with phenotype HbAA ABO blood type B+, while the patient was O+. Prior to HSCT, the patient underwent a MRI of the brain, including pseudocontinuous arterial spin labeling (pCASL) perfusion9 and oxygen extraction fraction (OEF)10 imaging. Pre-transplant, chronic left and right frontoparietal infarcts were noted, along with large areas of encephalomalacia in the middle cerebral artery distribution bilaterally. MR angiography was unchanged from prior studies. The patient’s hematocrit was 30%, with a moderately-elevated cortical CBF of 66.2 ml/100g/min in non-infarcted tissue and approximately normal OEF of 31.8%.
While engraftment was successful (HbAA genotype, 100% donor), the patient developed pure red cell aplasia (PRCA) related to the ABO incompatible HSCT, an autoimmune issue resulting in transfusion-dependent anemia. At this first post-transplant timepoint, the patient’s hematocrit was maintained at 25% with transfusion support. The patient’s CBF rose to 75.8 ml/100g/min with persistent anemia, and her OEF increased to 39.3%. No new infarcts were noted at this time, but cerebral hemodynamic measures remained concerning.
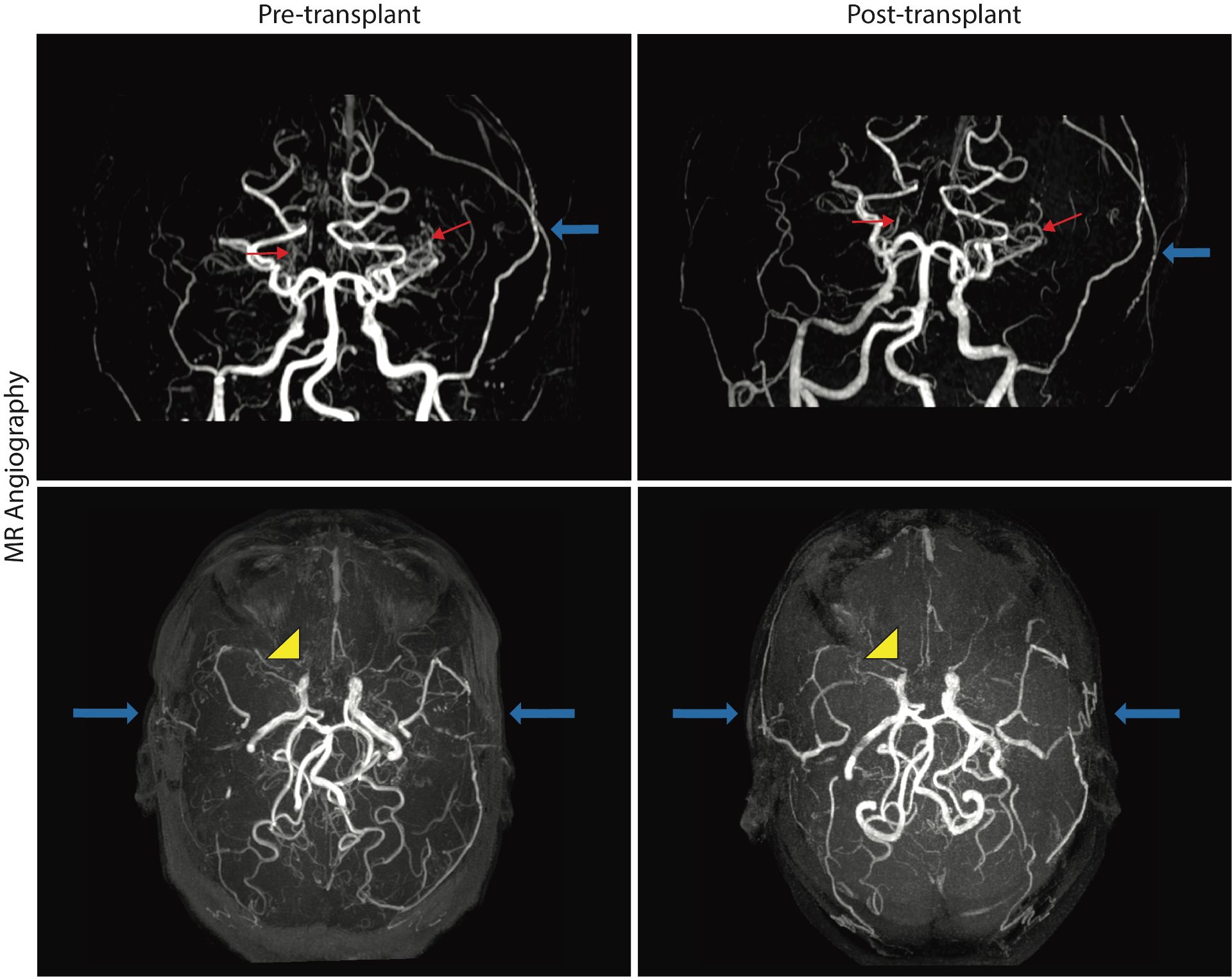
To address her PRCA and need for ongoing blood transfusions, the patient received two rituximab infusions, which were ineffective. She was subsequently given weekly injections of daratumumab 1800 mg for four weeks, which successfully treated her PRCA.11 Her hematocrit rose to 39%, within a normal range [36-48% in women],12 and has been maintained without transfusions. Correspondingly, CBF largely normalized to 38.2 ml/100g/min [normal range = 40-65 ml/100g/min],13,14,15 and the patient’s OEF was assessed at 36.3% [normal range = 35-45%].16 At a fourth timepoint, approximately three years post-transplant, cerebral hemodynamic measures remained stable, with hematocrit=41%, CBF=46.3 ml/100g/min, and OEF=40.4%. Overall, from pre-HSCT to post-HSCT after treatment for ABO incompatibility, the patient’s hematocrit increased, CBF normalized to an approximately healthy value (Figure 2), and OEF adjusted slightly, albeit within a normal range. No new strokes or silent cerebral infarcts have developed, and her cerebral vasculopathy remains severe but is stable at three years post-transplant (Figure 3).
Discussion
This case demonstrates that haploidentical HSCT was an effective treatment for mitigating impaired cerebral hemodynamics in a patient presenting with SCA and syndromic moyamoya and was not associated with new infarcts despite severe MMS with bilateral ICA occlusions.
Moreover, curative transplant has been shown to normalize CBF and OEF in both children6 and adults7,17 with SCD. However, because HSCT decreases gray matter CBF to approximately normal levels of 45-60 ml/100g/min, it is unclear whether decreasing CBF affects collateral flow pathways such as the lenticulostriate collaterals in the comorbid setting of MMS and places patients at risk for infarcts. More specifically, collateral flow trajectories are sensitively dependent on cerebral perfusion pressure, and it is logical that a therapy-induced change in CBF could detrimentally alter blood flow in these regions. Limited information is available on this possibility given that group-level neuroimaging studies of SCA may exclude patients with MMS or consider them only in sub-analysis given the competing effects of arterial steno-occlusion and anemia on CBF.
In this case, a patient with both SCA and MMS showed normalization of CBF and an OEF that maintained within an approximately normal range following HSCT. It is possible that HSCT, especially in conjunction with indirect revascularization surgery for MMS, may restore brain hemodynamics and contribute to stroke protection. Initially, development of pure red cell aplasia after transplant resulted in anemia with hematocrit levels similar to SCA without transplant. Hemodynamic impairment assessed by CBF was consistent with the level of anemia, rather than the hemoglobin phenotype, which changed from HbSS to HbAA after transplant. Following appropriate treatment for PRCA, the patient’s hemoglobin levels and CBF stabilized to within healthy physiological ranges, and she has not experienced any new cerebral infarctions with three years of post-transplant follow up.
Conclusion
This complex case of a patient with SCA and MMS who underwent HSCT, complicated by autoimmune pure red cell aplasia, demonstrates that HSCT can normalize cerebral hemodynamics without new stroke development, even as blood flow to collateral vessels decreases. Additionally, longitudinal functional neuroimaging performed before and after transplant effectively monitored cerebral hemodynamic changes. Following correction of severe anemia post-HSCT, normalization of CBF and hematocrit were observed, while OEF remained in an approximately normal range throughout treatment course. Curative transplants are often considered for patients at highest risk for cerebral infarction who may also have comorbid MMS. In this case, surveillance hemodynamic imaging supported the clinical benefit of HSCT in a patient with SCA and severe MMS, as demonstrated by normalization of CBF and lack of new infarct development. Although conclusions are limited by the nature of a single case report, these findings highlight the potential for successful response to HSCT and underscore the need for further study. Overall, this complex post-transplant trajectory illustrates the sensitivity of perfusion-weighted imaging methods in detecting cerebral hemodynamic changes after improvement in anemia following transplant.
Consent
Informed consent and assent were obtained from the patient’s guardian and the patient for this IRB-approved study.
Data Availability
The data that support the findings of this study are available on request from the corresponding author. The data are not publicly available due to privacy or ethical restrictions.
Conflicts of interest
Funding was provided as part of an investigator-initiated study supported by the National Institutes of Health. All other authors declare no conflicts of interest and have no relevant financial or non-financial interests to disclose.
Funding Statement
This work was supported in part by the NIH/NINDS (5R01NS123281), NIH/NIA (5R01AG062574), and NIH/NHLBI (1R01HL155207).
References
- Rees DC, Williams TN, Gladwin MT. Sickle-cell disease. Lancet. 2010;376(9757):2018-2031. doi:10.1016/S0140-6736(10)61029-X
- Schlotman AA, Donahue MJ, Kassim AA, et al. Intracranial and Extracranial Vascular Stenosis as Risk Factors for Stroke in Sickle Cell Disease. Pediatric Neurology. 2021;114:29-34. doi:10.1016/j.pediatrneurol.2020.10.006
- Scott RM, Smith ER. Moyamoya Disease and Moyamoya Syndrome. N Engl J Med. 2009;360(12):1226-1237. doi:10.1056/NEJMra0804622
- Jordan LC, DeBaun MR, Donahue MJ. Advances in neuroimaging to improve care in sickle cell disease. Lancet Neurol. 2021;20(5):398-408. doi:10.1016/S1474-4422(20)30490-7
- Kassim AA, de la Fuente J, Nur E, et al. An international learning collaborative phase 2 trial for haploidentical bone marrow transplant in sickle cell disease. Blood. 2024;143(25):2654-2665. doi:10.1182/blood.2023023301
- Hulbert ML, Fields ME, Guilliams KP, et al. Normalization of cerebral hemodynamics after hematopoietic stem cell transplant in children with sickle cell disease. Blood. 2023;141(4):335-344. doi:10.1182/blood.2022016618
- Aumann MA, Richerson W, Song AK, et al. Cerebral hemodynamic changes after haploidentical hematopoietic stem cell transplant in adults with sickle cell disease. Blood Adv. 2024;8(3):608-619. doi:10.1182/bloodadvances.2023010717
- Hulbert ML, McKinstry RC, Lacey JL, et al. Silent cerebral infarcts occur despite regular blood transfusion therapy after first strokes in children with sickle cell disease. Blood. 2011;117(3):772-779. doi:10.1182/blood-2010-01-261123
- Juttukonda MR, Jordan LC, Gindville MC, et al. Cerebral hemodynamics and pseudo-continuous arterial spin labeling considerations in adults with sickle cell anemia. NMR Biomed. 2017;30(2). doi:10.1002/nbm.3681
- Lu H, Ge Y. Quantitative evaluation of oxygenation in venous vessels using T2-Relaxation-Under-Spin-Tagging MRI. Magn Reson Med. 2008;60(2):357-363. doi:10.1002/mrm.21627
- Weverling F, Roeven M, Nijssen C, et al. Efficacy and safety of daratumumab in pure red cell aplasia after allogeneic transplantation: Dutch real-world data. Blood Advances. 2024;8(7):1683-1686. doi:10.1182/bloodadvances.2023011190
- Billett HH. Hemoglobin and Hematocrit. In: Walker HK, Hall WD, Hurst JW, eds. Clinical Methods: The History, Physical, and Laboratory Examinations. 3rd ed. Butterworths; 1990. Accessed September 10, 2025. http://www.ncbi.nlm.nih.gov/books/NBK259/
- Lewis CRA. Cerebral Blood Flow: Determinants. In: Freeman BS, Berger JS, eds. Anesthesiology Core Review: Part One Basic Exam. McGraw-Hill Education; 2014. Accessed September 10, 2025. accessanesthesiology.mhmedical.com/content.aspx?aid=1102568686
- Aumann MA, Richerson W, Song AK, et al. Cerebral hemodynamic changes after haploidentical hematopoietic stem cell transplant in adults with sickle cell disease. Blood Advances. 2024;8(3):608-619. doi:10.1182/bloodadvances.2023010717
- Jordan, Lori C., Richerson W, Aumann M, et al. Cerebral blood flow across the lifespan in sickle cell anemia. Neurology. In Press.
- Madsen LS, Thomsen MK, Angleys H, et al. Estimation of oxygen extraction fraction based on hemodynamic measurements using DSC-MRI. Imaging Neuroscience. 2025;3:imag_a_00562. doi:10.1162/imag_a_00562
- Afzali-Hashemi L, Dovern E, Baas KPA, et al. P1421: The Effect of Hematopoietic Stem Cell Transplantation on Cerebral Perfusion and Oxygen Metabolism in Patients with Sickle Cell Disease. HemaSphere. 2023;7(S3):e144919e. doi:10.1097/01.HS9.0000972572.14491.9e
Figure Legends
Figure 1. Timeline of patient imaging findings (left) and medical history (right). (MRI/MRA = magnetic resonance imaging/angiography, MCA = middle cerebral artery, ICA = internal carotid artery, ACA = anterior cerebral artery, MMS = moyamoya syndrome, Hb = hemoglobin).
Figure 2. Anatomical imaging of our patient with sickle cell anemia (HbSS) and moyamoya syndrome vasculopathy who was in her 20s at the time of imaging. Magnetic resonance images show chronic infarction (yellow arrows) throughout the right hemisphere on axial T1-weighted and T2-weighted FLAIR images at pre-transplant, post-transplant with ABO incompatibility (post-transplant 1) and post-transplant after successful treatment for pure red cell aplasia (post-transplant 2 and 3). At post-transplant 1, as an incidental finding, opacification of the frontal sinuses and mucosal thickening of the maxillary sinus is visible (white arrow). Bottom row: Perfusion maps show pre-transplant cerebral blood flow (CBF=66.2 ml/100g/min) is elevated secondary to anemia with hemoglobin level of 10.4 g/dL and increases at post-transplant 1 during ABO incompatibility (CBF=75.8 ml/100g/min, Hb=8.1 g/dL). However, CBF reduces to an approximately normal range outside of the infarct region at post-transplant 2 (CBF=38.2 ml/100g/min), with increased hemoglobin level of 13.3 g/dL, consistent with improved oxygen carrying capacity post-engraftment. At post-transplant 3, CBF remains stable within healthy physiological range (CBF=46.3 ml/100g/min), with a stable hemoglobin of 13.6 g/dL. See color bar on the right side of the figure, which shows CBF in ml/100g of brain tissue/minute.
Figure 3. Time of Flight magnetic resonance angiography (MRA) images at pre-transplant and three-years post-transplant with coronal (top row) and axial views (bottom row). MRAs demonstrate significant but stable lenticulostriate collateral formation (red arrows), bilateral surgical revascularization (blue arrows), and severe intracranial stenosis of the bilateral internal carotid arteries and proximal middle cerebral artery and anterior cerebral artery branches (yellow arrowheads on left side of image, stenoses are bilateral).
Hematopoietic Stem Cell Transplant