Review
Samantha Carey MD 1, Mubeen F. Rafay, MB.BS, FCPS, MSc. 2,3
1University of Manitoba, 2 Section of Pediatric Neurology, Department of Pediatrics and Child Health, University of Manitoba, 3Children’s Hospital Research Institute of Manitoba, Winnipeg, Manitoba, Canada
Corresponding Author
Mubeen F. Rafay
Associate Professor, Section of Pediatric Neurology
Department of Pediatric and Child Health, University of Manitoba
Children’s Hospital Research Institute of Manitoba
AE 308, 820 Sherbrook Street, Winnipeg, Manitoba, R3A 1R9
Tel # 1-204-787-2414, Fax # 1-204-787-1922
E-mail: mubeen.rafay@utoronto.ca
Copyright © belongs to author(s)
All rights reserved.
Any redistribution or reproduction of part or all of the contents in any form is prohibited other than the following:
- you may print or download to a local hard disk extracts for your personal and non-commercial use only
- you may copy the content to individual third parties for their personal use, but only if you acknowledge the website as the source of the material
You may not, except with our express written permission, distribute or commercially exploit the content. Nor may you transmit it or store it in any other website or other form of electronic retrieval system.
Abstract
Ischemic stroke involving exclusively the posterior circulation is infrequent in children. Although there is relative paucity of data regarding posterior than anterior circulation arterial ischemic stroke in children, significant differences have been observed in clinical presentation, etiological risk factors, and outcome among children with these two ischemic stroke subtypes. This review article is aimed at reviewing the current knowledge about the posterior circulation arterial ischemic stroke (posterior AIS) in children.
Introduction
Arterial ischemic stroke (AIS) is becoming an increasingly recognized occurrence in children. Population studies have shown that the rate of stroke expressed as number of children per year averages 2.5 – 3.3 per 100,000 children per year(1-4). With advancement in imaging modalities, it is argued that this is a conservative estimate of the true number of pediatric AIS(5). Over two-thirds of childhood AIS involve anterior circulation(1,2,6,7). Ischemic stroke involving exclusively the posterior circulation is infrequent in children, with a reported frequency ranging from 14.5% – 37% of all pediatric AIS(6-11). Incidence data exclusive to posterior AIS is relatively lacking, except for small population-based studies that estimated a mean crude incidence rate of 0.18 – 0.38 per 100,000 person-years(8,12). In addition, there was a paucity of pediatric data regarding the predisposing and etiological factors, treatment and prognosis of posterior AIS in children(8,9,13-15), until a recent large multicenter study, originating from the International Pediatric Stroke Study (IPSS) database, provided a detailed clinical description of posterior AIS in children(11). The study noted significant differences for clinical presentation, etiological risk factors, and outcome in children with posterior AIS when compared to anterior circulation ischemic strokes (anterior AIS)(11). Here we aim to review the current knowledge about posterior AIS in children.
Anatomy of Posterior Circulation and Vasculature
The posterior circulation is responsible for blood flow to the cerebellum, brain stem, midbrain, posterior cerebral cortex, and most of the thalamus. Vessels implicated in posterior circulation branch off the subclavian arteries and begin as paired vertebral arteries, one on each side. The vertebral arteries travel through the posterior arches of the cervical vertebrae to enter the skull through the foramen magnum. Intracranially, the paired vertebral arteries give rise to the posterior inferior cerebellar arteries just before fusing together to follow as the single basilar artery. The basilar artery first gives rise to the anterior inferior cerebellar arteries, followed by pontine branches and superior cerebellar arteries distally, prior to branching off as the paired posterior cerebral arteries. Each posterior cerebral artery joins the posterior communicating artery on the respective side to complete the circle of Willis along with anterior circulation. Previous studies have designated vertebral and posterior inferior cerebellar arteries as proximal, basilar artery as middle, and superior cerebellar artery, distal basilar tip and posterior cerebral artery as distal arteries. Each posterior cerebral artery supplies the posterior cerebral cortex (occipital lobe) and most of the thalamus on their respective side. The superior, anterior inferior cerebellar arteries and posterior inferior cerebellar arteries and other branches of vertebral arteries supply blood to the cerebellum, brain stem, and part of the spinal cord(16).
Etiologies and Risk Factors
Risk factors for ischemic stroke have been defined more adequately in adults as opposed to the pediatric population. In adults, atherosclerotic peripheral vascular and cardiac disease, hypertension, and diabetes mellitus are major causes of posterior AIS which are not seen in children. Risk factors that apply to both groups include cerebral arteriopathies (such as focal cerebral arteriopathy, traumatic or non-traumatic craniocervical arterial dissection, moyamoya), cardiac disease, hypercoagulable disorders, sickle cell disease, certain genetic conditions, increased intracranial pressure with brain herniation, and infectious and non-infectious cerebral vasculitis(1-3,17-21). In children with posterior AIS, identifiable risk factors are reported in over 68% of cases, with vasculopathy (mainly traumatic and non-traumatic vertebrobasilar arteriopathies), head and neck disorders and infection among the most common risk factors(7-11,14). As with anterior AIS, a significant proportion (11 – 32%) of posterior AIS cases are either idiopathic or of indeterminate etiology(7,8).
Cerebral arteriopathies are the most common cause of both posterior and anterior AIS in children, but more importantly, the presence of an arteriopathy is a strong predictor of AIS recurrence(20,22). Moyamoya, focal cerebral arteriopathy (idiopathic or post varicella associated focal arteriopathy) and craniocervical arterial dissection have all been linked to both initial stroke as well as stroke recurrence in children(20,23-29). Although most cerebral arteriopathies show predilection for anterior circulation (such as moyamoya, post varicella angiopathy), posterior AIS can also be associated with these predominantly anterior arteriopathies(11,20,26). Craniocervical arterial dissection (CCAD) is the most predominant arteriopathy associated with posterior rather than anterior AIS, in both children and young adults(9-11,14,26,30-33). Studies have shown that as many as 70% of children with AIS due to a vasculopathy have recurrent strokes, sharply contrasting to the complete lack of recurrent ischemic strokes in patients without preexisting vasculopathies(21,23,34).
Trauma is another important risk factor specific to posterior AIS, mainly due to its strong association with CCAD(30,31). Reportedly, CCAD accounts for 7.5-20% of all childhood AIS cases. Of these, over 70% of CCAD cases are due to extracranial dissection which compared to intracranial dissection is known to be more strongly associated with preceding trauma or congenital vascular or cervical spine abnormalities (1,6,9,20,30-32). Several studies cite that most patients with posterior AIS were boys with either vascular abnormalities or trauma leading to CCAD(9,30-32,35). In a study by Chabrier and colleagues, 4 out of 5 patients with cervical extracranial dissections experienced head and neck trauma whereas none of the seven patients with intracranial dissections experienced trauma in the ten days preceding the stroke(35). The incidence of preceding trauma has been variable across studies; in two separate studies, half of the posterior dissections were linked to some type of trauma, and other stroke etiologies that were not associated with preceding trauma(9,32). In the study by Rafay and colleagues, anterior circulation dissections had higher association with trauma, whereas posterior circulation dissections were more commonly spontaneous in children(32). A recent literature review of 16 articles with 182 CCAD cases indicated that CCAD is associated with head and/or neck trauma in 56% of AIS cases, and among those a majority (61%) presented with posterior AIS and extracranial vertebral artery involvement (60%)(31). A recent large multicenter study by the IPSS investigators found that CCAD was more common (20% versus 8.5%, p < 0.001) and more frequently associated with trauma (16% versus 8.5%, p < 0.001) with posterior AIS than anterior AIS(11).
Importantly, trauma does not need to be “major” to be associated with dissection. Up to 25% of the posterior arterial dissections are so called “spontaneous dissections” that either had no association with trauma or had trivial trauma reported at the time of presentation, indicating that the posterior extracranial vasculature is likely more susceptible to mild or trivial trauma(31,32). Reported risk factors among the spontaneous arterial dissection cases include congenital preexisting vertebrobasilar bony abnormalities (aberrant vertebral arcuate foramina, hereditary exostosis, etc.), certain genetic conditions (hyperhomocysteinemia, collagen vascular disease, Factor IX deficiency, trisomy 21, etc.), and athetoid cerebral palsy, indicating an underlying susceptibility to tear or dissection with trivial trauma(31). In addition, there are also rare reports of dynamic mechanical occlusion or narrowing of the vertebral during normal head and neck rotation or extension caused by the presence of abnormal bony structures such as fibrotendinous bands, bony spicules, tumor or disc herniation, resulting in symptoms of vertebrobasilar insufficiency, including evolution into posterior AIS. This has been reported in both children and adults and is referred to as Bow hunter’s syndrome or rotational vertebral artery occlusion syndrome, with one pediatric case report advocating for a specific imaging protocol to explore this rare mechanism(36-38). Based on the strong association between posterior AIS and head and neck trauma, current consensus recommendation is to consider head and neck trauma in all cases with posterior AIS in children and young adults, especially with CCAD, unless it is already known that traumatic dissection has occurred(1,31).
Genetic connective tissue disorders and collagen vascular and biochemical abnormalities with predisposition to thromboembolic events also account for a significant proportion of posterior AIS, either with or without arterial dissection(9,10,31,32,39,40). Monogenic connective tissue diseases reportedly associated with an increased risk for posterior AIS, include vascular form of Ehlers-Danlos syndrome (secondary to COL3A1 mutation), Marfan’s syndrome, Loeys-Dietz syndrome, pseudoxanthoma elasticum, and ICAM-1 and E4690 K gene polymorphisms. Other monogenic disorders associated with posterior AIS include fibromuscular dysplasia, α-1-antitrypsin deficiency, Fabry’s disease, late-onset Pompe disease, hyperhomocysteinemia, MTHFR 677T polymorphism, hereditary hemochromatosis, Osteogenesis imperfecta type 1, autosomal dominant polycystic kidney disease, and Turner and Williams syndromes. In addition, approximately 2-3% of familial cases have also been reported in the literature, with linkage studies implicating several collagen vascular genes (such as COL16A1, COL8A1, COL8A2, TIMP, PLOD, FBLN2) and a few chromosome loci (chromosome 15q24 and 10q26) (31,32,39,40).
A congenital cardiac abnormality as an underlying etiology is reported in up to 30% of children with pediatric AIS(1,7,41). It is generally speculated that cardioembolic stroke often involves posterior than anterior circulation, however the pediatric literature does not support this. In a cohort of 22 patients with posterior AIS, only 2 (9%) children had some form of a congenital cardiac disorder(31). In another pediatric study reviewing stroke recurrences among anterior and posterior AIS, 8 of the 46 (17.4%) posterior AIS cases had an underlying cardiogenic etiology(7). The large multicenter IPSS cohort of children with AIS demonstrated similar but statistically significant differences among anterior and posterior AIS cases and reported that only 19% of children with posterior AIS had cardioembolic strokes, compared to 32% with anterior AIS (p < 0.001)(11).
In both children and adults, infection and inflammation (especially involving head and neck) are risk factors for both anterior and posterior AIS, owing to the diffuse nature of the cerebral vasculature involvement(1,9,11,17,19).
Amongst hemoglobinopathies, sickle cell anemia is the most common risk factor for AIS in children. While generally sickle cell disease tends to spare the posterior circulation, severe sickling can lead to posterior AIS(42). Additionally, sickle cell disease often leads to intracranial vasculopathy, which further increases the risk of all stroke types in children(42,43)
Few other risk factors, such as diabetes mellitus, have been linked to increased risk for posterior AIS in adults. These may become applicable to children in the future, due to the increasing prevalence of type 2 diabetes and obesity in young adults(44,45). There is no data to support this in children at present.
Clinical Presentation
Children presenting with posterior AIS are more frequently males and older, compared to anterior AIS. In a European single center case series, all 22 cases with posterior AIS were non-neonates, and had a median age was 5.7 years, range 8 months to 16 years, and 77% were male(9). In an Australian single center study of 27 children with posterior AIS, the mean age at diagnosis was 7 years 11 months, and 70% were male(14). In a Canadian single center study of 158 children with AIS, 24 (15%) children, 22 non-neonates, mean age 5.3 years, 63% male, had a confirmed diagnosis of posterior AIS(8). In another larger North American single center series of 46 non-neonate children with posterior AIS, the median age was 9.2 years and 76% were male(7). The recent multicenter international pediatric study comparing posterior AIS to anterior found similar results and statistically confirmed that posterior AIS was more frequent than anterior AIS among non-neonates (22% versus 8.8%, p < 0.001), older children (median age 7.8 years versus 5.1 years, p < 0.001) and male sex (64 % versus 56%, p = 0.002)(11). Data on ethnicity for posterior AIS involvement is, however, limited and likely biased owing to under or over representation and reporting(8, 9,14).
Although awareness for pediatric stroke is increasing, due to the non-specific and atypical presentations it is still quite common for stroke diagnosis to be delayed or missed for many hours(46-48). Specific to posterior AIS, missed and delayed diagnosis is at least partly related to the non-specific and highly variable clinical presentation and extensive collateral circulation available in posterior head region(19,47,48).
Anterior strokes have more classic presentation symptoms that are more suspicious of AIS than posterior AIS presentation symptoms, which are often non-specific (19,31,49,50). Only a small proportion of patients with posterior AIS present with focal neurological deficits, such as visual field defects, ataxia, and hemisensory and motor symptoms(31,51). Most patients present with varying and often non-specific symptomatology, including ataxia, vertigo, headache, neck pain, and vomiting which are frequent presentation symptoms in other commonly seen neurological disorders (9,13,31,47,49,50,52). Few symptoms, such as head and neck pain, vomiting, and vertigo, particularly following a trauma, are considered strong indicators of posterior arterial dissection and resultant posterior AIS(9,30,31,35). In an adult study, by Libman and colleagues, it was noted that almost 20% of posterior AIS patients presented with vomiting, compared to just 3.5% of anterior strokes (p value <0.001)(49). In one study following children with posterior AIS, age range 2-17 years, 11 out of the 22 enrolled patients had pain in the neck and head regions(9). In another study, the majority of children with posterior dissection (5 out of 7) presented with warning symptoms compared to only one patient with anterior dissections (n=9). The most frequent warning symptoms among them included vomiting, neck pain, and headache(32). Clinical presentation with headache and vomiting combined with altered level of alertness usually points to increased intracranial pressure. Considering the crowded nature of the posterior fossa, raised intracranial pressure is not infrequent in children that have suffered posterior AIS(13,32). It is, therefore, advised that a patient presenting with this subset of symptoms should be thoroughly worked up for possibility of posterior circulation stroke (9,30-32,35).
In children with posterior AIS, the most common clinical presenting features have included impaired consciousness with or without abnormal posturing in over 70%, at least one sensorimotor deficit on assessment in over 65%, and a significant headache with or without neck pain in over 50% (8,9,11,14). Other presentations included speech disturbances (48%), ataxia (47%), visual field defects (41%) and vertigo (29%). Nausea, vomiting, vertigo, and mild headache preceding the stroke presentation are noted in 38 – 41% of posterior AIS cases(8,9,11,14). Compared to anterior AIS, headache was more common (54% versus 32%, p<0.001), whereas seizures were less frequent (19% versus 32%, p<0.001) in children with posterior AIS(11). As expected, unilateral versus bilateral motor deficits were common with anterior AIS, whereas other presentations are not different between anterior and posterior AIS(8,9,11,14). Although data is limited by reporting, median stroke severity at presentation measured by Pediatric NIH Stroke Scale (PedNIHSS) has been reportedly lower in posterior AIS than anterior AIS(8,9,11,14).
Radiological Presentation
Radiological data is a critical element in the diagnosis of posterior stroke. This is especially true in children, due to the breadth of potential diagnoses available for a child with focal or general neurological deficits and non-specific symptomatology(47,53,54).
Head CT scan, while widely available and still readily used, does not allow adequate visualization of posterior head regions and hence MRI brain is the best neuroimaging modality for the posterior fossa(5,14,47). In addition, as with anterior circulation strokes, head CT may not show parenchymal changes for several hours following an infarct (47,48). Early parenchymal changes that appear in acute ischemic stroke are more readily identified by brain MRI than by CT, contrasted with hemorrhagic strokes, where head CT is the best modality for its detection. Diffusion weight imaging (DWI) MRI sequences can demonstrate an infarct within 5-10 minutes of ischemic event. Therefore, MRI brain is currently considered the most appropriate first line imaging modality for identifying ischemic stroke, including posterior AIS in pediatric population(1,53,55) (Figure). This interval is maintained at maximum intensity for 4-7 days and sometimes lasts up to 10 days, which helps confirm stroke event being acute or sub-acute in nature. In the recent multicenter posterior AIS study, IPSS investigators showed that the CT brain was far inferior to MRI brain as an initial imaging modality in the diagnosis of posterior AIS (CT brain 32% versus MRI brain 68%) than to anterior AIS (CT 44%, versus MRI brain 56%) (11). Furthermore. in pediatric studies, distal infarcts have been shown to be more common than proximal infarcts with posterior AIS(9,14).
With regards to vascular imaging, advancements in computed tomography angiography (CTA) and magnetic resonance angiography (MRA) have allowed vascular abnormalities to be visualized readily in a non-invasive manner (Figure). Since arteriopathies including dissection, stenosis, embolism, and other irregularities are a major cause of posterior AIS and its recurrence(8,9,11,13,30,32,35), non-invasive vascular imaging is recommended for all children with all types of AIS(1, 53). In addition, if dissection is suspected, some specific MRI brain sequences, such as axial T2 fat saturated images, are helpful in identifying arterial wall abnormalities such as the crescent sign, presence of double lumen, intimal flap, pseudoaneurysm, or vessel occlusion/stenosis(1,31,32). While non-invasive techniques are improving in quality, they may still present false negatives, and may not be adequate for all intraluminal arterial abnormalities (such as seen with inflammation and arterial dissection)(1,31,32,35). In patients where such abnormalities are clinically suspected and non-invasive vascular imaging is nonrevealing (such as with history of trauma) conventional cerebral catheter angiography remains the most definitive diagnostic vascular imaging modality(1,22,30,31). In addition, it may also be warranted when a subtle or unclear abnormality is detected by cerebral MRA and CTA since these studies are known to overestimate or underestimate the extent of the visualized abnormality(1,31). In one study of pediatric dissection, all patients required conventional angiography to clinch the diagnosis of dissection(32).
Despite advances, neuroimaging for posterior AIS remains challenging as sensitivity and specificity of MRI/MRA is relatively lower in vertebral arteries compared to carotids, even despite targeted studies improving the yield somewhat(9,14,31,32). Half of the posterior circulation strokes found in one study were due to dissection, but this was only discovered with vascular invasive imaging(9). This is thought to be a conservative estimate, since many of the other patients’ imaging studies did not include cerebral catheter angiography and could not conclusively prove that dissection was not the cause of stroke in the posterior circulation(9,14).
Treatment
Treating pediatric AIS continues to be challenging and controversial. Current approaches are largely based on data from adult stroke studies(1,19,56). Although most treatment approaches are supported by large pediatric case-based studies(2,18,57), no randomized controlled trials are available in pediatric stroke literature, except in sickle cell population(58). This explains the variations in pediatric stroke treatment guidelines across centers and why no definitive treatment plans have been established to date (1,55,56, 59-61). Severity of neurological sequelae has been clearly outlined and therefore further investigation into more aggressive and targeted treatment is warranted(2,62,63). As we gain more published experience in the use of antithrombotic and fibrinolytic agents in the treatment of pediatric AIS, we are becoming more sure that these agents are safe to use in the pediatric population, however questions still remain regarding their efficacy and optimal dose(1,56,57,64-66).
Recent good quality pediatric data has demonstrated the feasibility and safety of systemic thrombolysis with intravenous tissue plasminogen activator (tPA) if administered within 4.5 hours of stroke symptom onset in pediatric AIS population(1,55,59,60,64,65). More recently, endovascular recanalization procedure (such as mechanical endovascular thrombectomy) is shown to be of comparable efficacy and safety in children with AIS as in adults with AIS, if done typically within 6 hours and up to 24 hours in select cases(67). In the literature, both adults and children with posterior AIS due to basilar artery occlusion have been treated beyond 20 hours(67-69). In one case report, a child with brainstem stroke secondary to a basilar artery occlusion was treated successfully at least 20 hours after stroke symptom onset(69). Adult studies show concordant evidence with one review stating 52% of posterior AIS patients achieved reperfusion using alteplase (tPA) 20 hours after symptom onset(70). There are, however, many factors limiting the ability to use thrombolytics in acute arterial ischemic stroke in children, but one of the most likely reasons use of thrombolytics in childhood stroke is understudied is due to the delay in recognition of stroke in children compared to adults(47,48). Other major barriers include differences in presentation, coagulation systems, infrequency of pediatric stroke occurrence, and lack of organized acute pediatric stroke care at most institutions caring for these children(56,64,65,71).
American Heart Association adult stroke guidelines recommend use of intravenous tPA as a fibrinolytic for hyperacute stroke treatment (Class I, Level of Evidence A)(72), which cannot be extrapolated to pediatric populations due to differences in risk factors, recovery and optimal dose and also due to lack of randomized data for efficacy of this agent in children with ischemic stroke. Due to the lack of rigorous evidence, use of hyperacute stroke treatment in children with stroke is currently not standard of care by the American Heart Association, Canadian Stroke Consortium, United Kingdom Royal College of Physicians, Australian and the American College of Chest Physicians pediatric stroke management guidelines(1,55,59,60). However, all pediatric stroke guidelines are supportive of hyperacute treatment with systemic tPA and or mechanical thrombectomy procedure for children with AIS due to large vessel occlusion, provided certain conditions are met and their use is limited to centers with pediatric stroke expertise and protocols in place or as part of research trials(1).
For both secondary stroke prevention, almost all have recommended initial antiplatelet therapy with aspirin(1,55,59,60,73). Some cases with high stroke recurrence risk or multiple stroke recurrences warrant use of anticoagulation, namely AIS secondary to arterial dissections, cardioembolic strokes and certain hypercoagulable states disorders(1). Interestingly, published literature indicates that children with posterior AIS during the acute presentation more often received an antithrombotic therapy compared to children with anterior AIS (84% versus 77.1%, p < 0.003)(11). While to date it has not yet been shown in children that use of anticoagulant agent (such as heparin, enoxaparin) have prognostic benefit for posterior circulation strokes compared to antiplatelet agent, current practice is to use these agents in certain types of posterior AIS(1,55,60). Interestingly, pediatric studies show that the number needed to treat to prevent recurrence using heparin is 10, which is low compared to number needed to treat of 135 for aspirin(56). Despite this, use of aspirin is still prevalent in children with stroke, and treatment regimens vary widely depending on type of stroke and risk factors(52,56). Adult trials have shown risk reduction of 7 per 1000 in the aspirin group, with risk of hemorrhage increasing only to 2 per 1000(74). No pediatric-specific trials have been done, but all recently published pediatric stroke management guidelines have extrapolated from the adult recommendations. In children with AIS, antiplatelet therapy with aspirin, dosages of 3-5 mg/kg/day, is recommended as prophylactic therapy, unless there are other contraindications or specific reasons to consider anticoagulant therapy (1,55,59,60,73).
As in adults, for children with AIS, current published guidelines also strongly recommend treatment and monitoring for fever, seizures, hyper and hypoglycemia, and raised intracranial pressure. It is not uncommon for posterior AIS patients, even with moderate infarct size, to manifest increased intracranial pressure often limited to the posterior fossa because of the already crowded nature of this region. It is, therefore, imperative to monitor patients with posterior AIS more carefully(1,55,59,60,73). In addition, posterior AIS is known to recur frequently in the acute period thereby requiring close monitoring(9,13).
Despite known differences in pathophysiology, treatment of pediatric AIS still follows an adult model of antithrombotic therapy, intensive care support, and post stroke age-appropriate rehabilitation including school placement and supports, due to the present relative lack of direct evidence for various stroke treatments in the pediatric population.
Recurrence
General rate of stroke recurrence for all ischemic strokes in children is between 8.2% – 30%. Posterior AIS reportedly has a significantly higher risk of recurrence in the first 12 months compared to other territories(7,11,21,22,51,63,75). In the European single-center study specifically looking at recurrences in children with posterior AIS, a significantly high recurrence rate of 50% was noted (4 AIS and 7 TIA’s in 22 patients, with a median time to recurrence of one week)(9). Similar data was reported by the Australian study, finding that 52% of patients with posterior AIS had recurrent strokes(14). In the Australian paper, it was noted that about half of recurrences involved posterior and the other half affected the anterior circulation(14). Subsequent studies, however, indicated a relatively lower recurrence risk(10,12). In a small single center North American case series reviewing children with posterior AIS, 5 out of the 16 surviving children (31%) had ischemic stroke recurrence(8). Two studies specifically compared stroke recurrence rates among anterior and posterior AIS. A large single-center North American case series reviewed 107 pediatric AIS cases and found that 19% of posterior AIS cases (9 out of 46) had an ischemic stroke recurrence compared to 4% with anterior AIS (2 out of 61)(7). The most recent largest multicenter study based on IPSS database confirmed these findings as statistically significant differences in rate of stroke recurrence were identified among posterior AIS cases compared with anterior AIS cases {30% (66/217) versus 22% (115/522), OR 1.69, p = 0.02}(11).
Furthermore, etiology of the stroke has a clear role in recurrence; heart conditions,
coagulopathies, or vasculopathies that are untreated will more likely result in recurrence of stroke compared to children who were previously healthy at stroke onset (17,18,22,63). One study found that non-abrupt stroke onset was positively correlated to arteriopathies(52). They also found that all of the posterior AIS patients in the study (n=3) had non-abrupt stroke onset, which may mean that the posterior AIS were more often associated with vasculopathy, as well as had a high risk of stroke recurrence(52). In a study investigating craniocervical arterial dissection and risk of stroke recurrence rate(32), seven posterior circulation strokes among the 24 arterial dissection cases, two patients had a recurrent posterior AIS event after the initial dissection. This contrasted to none of the anterior dissection patients having recurrent events. Reasons for this may relate to the increased incidence of extracranial dissection in posterior circulation, and increased susceptibility to minor trauma in this circulation(32). Recent data also confirms that recurrent ischemic events are indeed more frequent with CCAD. Uohara and colleagues found that 5 out of 19 cases (26.3%) with vertebrobasilar dissection(7). Barbara and colleagues estimated an odds ratio of 2.39 for stroke recurrence among craniocervical arterial dissection cases (95% CI 1.36 – 4.22; p=0.003) (11).
The above data clearly indicates that recurrence is a major concern that requires close follow-up after acute presentation of a pediatric stroke involving the posterior circulation.
Outcome
Overall, studies have shown that at least half of the AIS child survivors have neurological sequelae (sensorimotor impairments, cognitive and behavioral difficulties, or seizures) (2, 62,63,75,76). In one Canadian population-based study, statistically significant predictors of outcome were outlined using the pediatric stroke outcome measure (PSOM) developed for that study, and different presentation characteristics(62). In general, the outcome of AIS in children is linked to age at index event, initial stroke severity, delay in stroke diagnosis, underlying cause, presence of an associated neurological disorder, and size of infarct and stroke recurrence risk(18,21,62,75,76), but such systematic outcome data exclusive to posterior AIS is limited.
The Pediatric National Institute of Health Stroke Severity Scale (PedNIHSS) is used frequently as a prognostic factor in pediatric AIS of any etiology and location(8, 41). Despite this, there has been some controversy in the efficacy of the National Institute of Health’s Stroke Severity Scale in determining severity of different types of strokes. It is proposed that the NIHSS places heavier weight on anterior circulation symptoms such as cortical signs and motor function, compared to posterior symptoms, like ataxia(50). In adult studies, posterior AIS have been shown to have lower scores on the NIHSS and better outcomes at 3 months(49). Although not studied in large multicenter pediatric stroke outcome studies(2,9,63), one small pediatric case series found similar association, and correlated a high PedNIHSS score with poor outcome in children with posterior AIS(8). In addition, many pediatric studies indicate that delay in stroke leads to delay in treatment, which in turn can result in worse outcomes(46-48). In one pediatric study, posterior AIS patients had increased time to diagnosis(47). Although initial stroke severity, diagnostic delay, recurrence risk, stroke etiology, and infarct size are all identified as useful determinants of outcome with childhood posterior AIS(7-9), at present we have limited concrete data on which is the most important of these factors, and which other factors may contribute as well.
In children with posterior AIS, moderate to severe neurological sequalae have been reported in at least 45% of posterior AIS survivors and death ranging from 2.7% to 26% of cases(7-11,14). In the European single center study, 12 of the 22 posterior AIS cases were unimpaired, and 2 cases only had mild residual deficits after a follow-up period of 4 years, meaning 63% of patients had a favourable outcome. The residual deficits in their cohort consisted of hemiparesis, hemianopia, ataxia, and cortical visual impairment(9). In the Canadian pediatric single center population-based study of posterior AIS, 14 of the 21 children had 24 months outcome data available (5 children died with index stroke presentation, one died several months later due to aspiration pneumonia and one lost to follow-up). Mean PSOM score was 1.7, with PSOM < 2 in 55% (8/14), including no impairment in 21% (3/14) of their cohort(8). The recent multicenter IPSS study demonstrated similar results and found residual neurological sequalae in 60% of posterior AIS survivors. They also found that compared to non-neonatal anterior AIS survivors, posterior AIS survivors had better outcomes after a median follow-up duration of 1.5 years; normal neurologic examination at the time of hospital discharge (29% versus 21%, p=0.002), less need for help, and low median PSOM score (33% versus 44%, p=0.03). However, death rate was similar at 2.9% in both anterior and posterior AIS cases(11).
Area for Further Research
Posterior circulation AIS is infrequent in the pediatric population and, as such, is understudied at present. As outlined above, certain etiologies and risk factors carry a strong association with posterior AIS, and survivors of posterior AIS appears to have better outcomes than anterior AIS. More investigation needs to be done to determine the most effective and safe antithrombotic treatment, optimal management of primary preventative measures and rehabilitation and prognostic factors following posterior stroke to maximize the potential for improvement in this population where improvement and resolution of deficits is increasingly likely. Finally, more research needs to be dedicated towards idiopathic stroke of unknown etiology, specifically which risk factors require primary preventative measures. Challenges arise because randomized controlled trials are often not feasible in the pediatric population. As such, continuation of large multicenter case studies and a meta-analysis of multiple studies could have the potential to help target an optimal treatment and clinical outcomes, but this requires systematic research approaches utilizing standardized outcome measures, both clinical and patient reported outcomes, across studies.
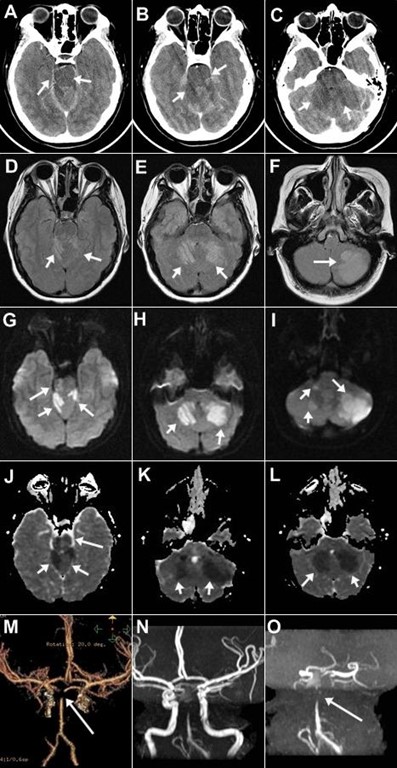
Figure Legend
15-year-old right-handed previously healthy girl presented with dizziness, ataxia, and swaying to one side. Head CT (A, B, C) showing hypointenisties involving pons and left and right cerebellar hemisphere suspicious for acute ischemic stroke. MRI brain FLAIR sequence (D, E, F) demonstrating multiple hyperintense signal abnormalities and DWI (G, H, I) and ADC sequences (J, K, L) showing areas of restricted diffusion with a corresponding apparent diffusion coefficient map involving the right superior cerebellar cortex, left cerebellar cortex, dentate nuclei and within the right anterolateral aspect of the pons, consistent with acute ischemic infarction involving these regions. CT angiogram (M) and time of flight MR angiogram (N, O) demonstrating lack of flow in the superior portion of the basilar artery, consistent with a basilar tip thrombosis. The right and left vertebral arteries appear patent and normal.
References
(1) Ferriero DM, Fullerton HJ, Bernard TJ, et al. Management of Stroke in Neonates and Children: A Scientific Statement From the American Heart Association/American Stroke Association. Stroke 2019 Mar;50:e51-e96.
(2) DeVeber G, Kirton A, Booth FA, et al. Epidemiology and Outcomes of Arterial Ischemic Stroke in Children: the CanadianPediatric Ischemic Stroke Registry. Pediatr Neurol 2017;69:58-70.
(3) Giroud M, Lemesle M, Gouyon JB, Nivelon JL, Milan C, Dumas R. Cerebrovascular disease in children under 16 years of age in the city of Dijon, France: a study of incidence and clinical features from 1985 to 1993. J Clin Epidemiol 1995 Nov;48:1343-1348.
(4) Lynch JK, Hirtz DG, deVeber G, Nelson KB. Report of the National Institute of Neurological Disorders and Stroke workshop on perinatal and childhood stroke. Pediatrics 2002 Jan;109:116-123.
(5) Agrawal N, Johnston SC, Wu YW, Sidney S, Fullerton HJ. Imaging data reveal a higher pediatric stroke incidence than prior US estimates. Stroke 2009 Nov;40:3415-3421.
(6) Mackay MT, Wiznitzer M, Benedict SL, Lee KJ, deVeber GA, Ganesan V. Arterial ischemic stroke risk factors: the International Pediatric Stroke Study. Ann Neurol 2011 Jan;69:130-140.
(7) Uohara MY, Beslow LA, Billinghurst L, et al. Incidence of Recurrence in Posterior Circulation Childhood Arterial Ischemic Stroke. JAMA Neurol 2017 Mar 1;74:316-323.
(8) Carey S, Wrogemann J, Booth FA, Rafay MF. Epidemiology, Clinical Presentation, and Prognosis of Posterior Circulation Ischemic Stroke in Children. Pediatr Neurol 2017 Sep;74:41-50.
(9) Ganesan V, Chong WK, Cox TC, Chawda SJ, Prengler M, Kirkham FJ. Posterior circulation stroke in childhood: risk factors and recurrence. Neurology 2002 Nov 26;59:1552-1556.
(10) Mccrea N | Saunders D | Bagkeri E | Chitre M | Ganesan V. Diagnosis of vertebral artery dissection in childhood posterior circulation arterial ischaemic stroke., 58 ed 2016:63-70.
(11) Goeggel SB, Rafay MF, Chung M, et al. Comparative study of posterior and anterior circulation stroke in childhood: Results from the International Pediatric Stroke Study. Neurology 2020 Jan 28;94:e337-e344.
(12) Fink M, Slavova N, Grunt S, et al. Posterior Arterial Ischemic Stroke in Childhood. Stroke 2019 Sep;50:2329-2335.
(13) Frosk P, Salman MS, Wrogemann J, Shah N, Rafay MF. Recurrent posterior circulation stroke in an infant with basilar artery aneurysm. J Child Neurol 2009 Aug;24:1019-1020.
(14) Mackay MT, Prabhu SP, Coleman L. Childhood posterior circulation arterial ischemic stroke. Stroke 2010 Oct;41:2201-2209.
(15) van der Aa NE, Dudink J, Benders MJ, et al. Neonatal posterior cerebral artery stroke: clinical presentation, MRI findings, and outcome. Dev Med Child Neurol 2013 Mar;55:283-290.
(16) The Blood Supply of the Brain and Spinal Cord. Sunderland (MA): Sinauer Associates; 2001.
(17) Fullerton HJ, Wu YW, Zhao S, Johnston SC. Risk of stroke in children: ethnic and gender disparities. Neurology 2003 Jul 22;61:189-194.
(18) Goldenberg NA, Bernard TJ, Fullerton HJ, Gordon A, DeVeber G. Antithrombotic treatments, outcomes, and prognostic factors in acute childhood-onset arterial ischaemic stroke: a multicentre, observational, cohort study. Lancet Neurol 2009 Dec;8:1120-1127.
(19) Rafay MF. Ischaemic stroke in children–overview including an Asian perspective. J Pak Med Assoc 2008 Jul;58:384-390.
(20) Wintermark M, Hills NK, deVeber GA, et al. Arteriopathy diagnosis in childhood arterial ischemic stroke: results of the vascular effects of infection in pediatric stroke study. Stroke 2014 Dec;45:3597-3605.
(21) Lanthier S, Carmant L, David M, Larbrisseau A, de VG. Stroke in children: the coexistence of multiple risk factors predicts poor outcome. Neurology 2000 Jan 25;54:371-378.
(22) Fullerton HJ, Wu YW, Sidney S, Johnston SC. Risk of recurrent childhood arterial ischemic stroke in a population-based cohort: the importance of cerebrovascular imaging. Pediatrics 2007 Mar;119:495-501.
(23) Braun KP, Bulder MM, Chabrier S, et al. The course and outcome of unilateral intracranial arteriopathy in 79 children with ischaemic stroke. Brain 2009 Feb;132:544-557.
(24) Chabrier S, Sebire G, Fluss J. Transient Cerebral Arteriopathy, Postvaricella Arteriopathy, and Focal Cerebral Arteriopathy or the Unique Susceptibility of the M1 Segment in Children With Stroke. Stroke 2016 Oct;47:2439-2441.
(25) Fullerton HJ, Stence N, Hills NK, et al. Focal Cerebral Arteriopathy of Childhood: Novel Severity Score and Natural History. Stroke 2018 Nov;49:2590-2596.
(26) Rafay MF, Shapiro KA, Surmava AM, et al. Spectrum of cerebral arteriopathies in children with arterial ischemic stroke. Neurology 2020 Jun 9;94:e2479-e2490.
(27) Sebire G, Meyer L, Chabrier S. Varicella as a risk factor for cerebral infarction in childhood: a case-control study. Ann Neurol 1999 May;45:679-680.
(28) Sebire G. Transient cerebral arteriopathy in childhood. Lancet 2006 Jul 1;368:8-10.
(29) Smith ER, Scott RM. Moyamoya: epidemiology, presentation, and diagnosis. Neurosurg Clin N Am 2010 Jul;21:543-551.
(30) Fullerton HJ, Johnston SC, Smith WS. Arterial dissection and stroke in children. Neurology 2001 Oct 9;57:1155-1160.
(31) Nash M, Rafay MF. Craniocervical Arterial Dissection in Children: Pathophysiology and Management. Pediatr Neurol 2019 Jun;95:9-18.
(32) Rafay MF, Armstrong D, DeVeber G, Domi T, Chan A, MacGregor DL. Craniocervical arterial dissection in children: clinical and radiographic presentation and outcome. J Child Neurol 2006 Jan;21:8-16.
(33) Schievink WI, Mokri B, Piepgras DG. Spontaneous dissections of cervicocephalic arteries in childhood and adolescence. Neurology 1994 Sep;44:1607-1612.
(34) Fullerton HJ, Wintermark M, Hills NK, et al. Risk of Recurrent Arterial Ischemic Stroke in Childhood: A Prospective International Study. Stroke 2016 Jan;47:53-59.
(35) Chabrier S, Lasjaunias P, Husson B, Landrieu P, Tardieu M. Ischaemic stroke from dissection of the craniocervical arteries in childhood: report of 12 patients. Eur J Paediatr Neurol 2003;7:39-42.
(36) Braga BP, Sillero R, Pereira RM, et al. Dynamic compression in vertebral artery dissection in children: apropos of a new protocol. Childs Nerv Syst 2020 Nov 6.
(37) Rollins N, Braga B, Hogge A, Beavers S, Dowling M. Dynamic Arterial Compression in Pediatric Vertebral Arterial Dissection. Stroke 2017 Apr;48:1070-1073.
(38) Duan G, Xu J, Shi J, Cao Y. Advances in the Pathogenesis, Diagnosis and Treatment of Bow Hunter’s Syndrome: A Comprehensive Review of the Literature. Interv Neurol 2016 Jun;5:29-38.
(39) Grond-Ginsbach C, Debette S. The association of connective tissue disorders with cervical artery dissections. Curr Mol Med 2009 Mar;9:210-214.
(40) Debette S, Markus HS. The genetics of cervical artery dissection: a systematic review. Stroke 2009 Jun;40:e459-e466.
(41) Ziesmann MT, Nash M, Booth FA, Rafay MF. Cardioembolic stroke in children: a clinical presentation and outcome study. Pediatr Neurol 2014 Oct;51:494-502.
(42) Steen RG, Emudianughe T, Hankins GM, et al. Brain imaging findings in pediatric patients with sickle cell disease. Radiology 2003 Jul;228:216-225.
(43) Pegelow CH, Macklin EA, Moser FG, et al. Longitudinal changes in brain magnetic resonance imaging findings in children with sickle cell disease. Blood 2002 Apr 15;99:3014-3018.
(44) Nathan BM, Moran A. Metabolic complications of obesity in childhood and adolescence: more than just diabetes. Curr Opin Endocrinol Diabetes Obes 2008 Feb;15:21-29.
(45) Subramanian G, Silva J, Silver FL, et al. Risk factors for posterior compared to anterior ischemic stroke: an observational study of the Registry of the Canadian Stroke Network. Neuroepidemiology 2009;33:12-16.
(46) Gabis LV, Yangala R, Lenn NJ. Time lag to diagnosis of stroke in children. Pediatrics 2002 Nov;110:924-928.
(47) Rafay MF, Pontigon AM, Chiang J, et al. Delay to diagnosis in acute pediatric arterial ischemic stroke. Stroke 2009 Jan;40:58-64.
(48) Srinivasan J, Miller SP, Phan TG, Mackay MT. Delayed recognition of initial stroke in children: need for increased awareness. Pediatrics 2009 Aug;124:e227-e234.
(49) Libman RB, Kwiatkowski TG, Hansen MD, Clarke WR, Woolson RF, Adams HP. Differences between anterior and posterior circulation stroke in TOAST. Cerebrovasc Dis 2001;11:311-316.
(50) Sato S, Toyoda K, Uehara T, et al. Baseline NIH Stroke Scale Score predicting outcome in anterior and posterior circulation strokes. Neurology 2008 Jun 10;70:2371-2377.
(51) Bamford J, Sandercock P, Dennis M, Burn J, Warlow C. Classification and natural history of clinically identifiable subtypes of cerebral infarction. Lancet 1991 Jun 22;337:1521-1526.
(52) Braun KP, Rafay MF, Uiterwaal CS, Pontigon AM, DeVeber G. Mode of onset predicts etiological diagnosis of arterial ischemic stroke in children. Stroke 2007 Feb;38:298-302.
(53) Mirsky DM, Beslow LA, Amlie-Lefond C, et al. Pathways for Neuroimaging of Childhood Stroke. Pediatr Neurol 2017 Apr;69:11-23.
(54) Shellhaas RA, Smith SE, O’Tool E, Licht DJ, Ichord RN. Mimics of childhood stroke: characteristics of a prospective cohort. Pediatrics 2006 Aug;118:704-709.
(55) Medley TL, Miteff C, Andrews I, et al. Australian Clinical Consensus Guideline: The diagnosis and acute management of childhood stroke. Int J Stroke 2019 Jan;14:94-106.
(56) Bernard TJ, Goldenberg NA, Armstrong-Wells J, Amlie-Lefond C, Fullerton HJ. Treatment of childhood arterial ischemic stroke. Ann Neurol 2008 Jun;63:679-696.
(57) Schechter T, Kirton A, Laughlin S, et al. Safety of anticoagulants in children with arterial ischemic stroke. Blood 2012 Jan 26;119:949-956.
(58) Adams RJ, Brambilla DJ, Granger S, et al. Stroke and conversion to high risk in children screened with transcranial Doppler ultrasound during the STOP study. Blood 2004 May 15;103:3689-3694.
(59) Monagle P, Chan AKC, Goldenberg NA, et al. Antithrombotic therapy in neonates and children: Antithrombotic Therapy and Prevention of Thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest 2012 Feb;141:e737S-e801S.
(60) Paediatric Stroke Working Group, 2004 UK: Royal College of Physicians. Stroke in Childhood: Clinical Guidelines for Diagnosis,Management and Rehabilitation. Clinical Effectiveness and Evaluation Unit. 2004.
(61) Rafay MF. Paediatric stroke management guidelines. J Pak Med Assoc 2010 Mar;60:161-162.
(62) deVeber GA, MacGregor D, Curtis R, Mayank S. Neurologic outcome in survivors of childhood arterial ischemic stroke and sinovenous thrombosis. J Child Neurol 2000 May;15:316-324.
(63) Felling RJ, Rafay MF, Bernard TJ, et al. Predicting Recovery and Outcome after Pediatric Stroke: Results from the International Pediatric Stroke Study. Ann Neurol 2020 Jun;87:840-852.
(64) Amlie-Lefond C, Chan AK, Kirton A, et al. Thrombolysis in acute childhood stroke: design and challenges of the thrombolysis in pediatric stroke clinical trial. Neuroepidemiology 2009;32:279-286.
(65) Amlie-Lefond C, DeVeber G, Chan AK, et al. Use of alteplase in childhood arterial ischaemic stroke: a multicentre, observational, cohort study. Lancet Neurol 2009 Jun;8:530-536.
(66) Amlie-Lefond C, Shaw DWW, Cooper A, et al. Risk of Intracranial Hemorrhage Following Intravenous tPA (Tissue-Type Plasminogen Activator) for Acute Stroke Is Low in Children. Stroke 2020 Feb;51:542-548.
(67) Sporns PB, Strater R, Minnerup J, et al. Feasibility, Safety, and Outcome of Endovascular Recanalization in Childhood Stroke: The Save ChildS Study. JAMA Neurol 2020 Jan 1;77:25-34.
(68) Kaesmacher J, Maamari B, Meinel TR, et al. Effect of Pre- and In-Hospital Delay on Reperfusion in Acute Ischemic Stroke Mechanical Thrombectomy. Stroke 2020 Oct;51:2934-2942.
(69) Kirton A, Wong JH, Mah J, et al. Successful endovascular therapy for acute basilar thrombosis in an adolescent. Pediatrics 2003 Sep;112:e248-e251.
(70) MacLeod M. Current issues in the treatment of acute posterior circulation stroke. CNS Drugs 2006;20:611-621.
(71) Amlie-Lefond C, Wainwright MS. Organizing for Acute Arterial Ischemic Stroke in Children. Stroke 2019 Dec;50:3662-3668.
(72) Powers WJ, Rabinstein AA, Ackerson T, et al. 2018 Guidelines for the Early Management of Patients With Acute Ischemic Stroke: A Guideline for Healthcare Professionals From the American Heart Association/American Stroke Association. Stroke 2018 Mar;49:e46-e110.
(73) Wein T, Lindsay MP, Cote R, et al. Canadian stroke best practice recommendations: Secondary prevention of stroke, sixth edition practice guidelines, update 2017. Int J Stroke 2018 Jun;13:420-443.
(74) Chen ZM, Sandercock P, Pan HC, et al. Indications for early aspirin use in acute ischemic stroke : A combined analysis of 40 000 randomized patients from the chinese acute stroke trial and the international stroke trial. On behalf of the CAST and IST collaborative groups. Stroke 2000 Jun;31:1240-1249.
(75) Chabrier S, Husson B, Lasjaunias P, Landrieu P, Tardieu M. Stroke in childhood: outcome and recurrence risk by mechanism in 59 patients. J Child Neurol 2000 May;15:290-294.
(76) Ganesan V, Hogan A, Shack N, Gordon A, Isaacs E, Kirkham FJ. Outcome after ischaemic stroke in childhood. Dev Med Child Neurol 2000 Jul;42:455-461.
Posterior Circulation Stroke in Children
