Association of Plasma Inflammatory Proteins and Intracerebral Hemorrhage in Pediatric Brain Arteriovenous Malformations
Shweta A. Chawla, MS (1); Shantel Weinsheimer, PhD (1); Jeffrey Nelson, MS (1); Charles E. McCulloch, PhD (2); Ethan Winkler, MD, PhD (3); Nalin Gupta, MD (3); Daniel Cooke, MD (4); Steven Hetts, MD (4); Yi Li, MD (4); Christine K. Fox, MD, MAS (5); Heather J. Fullerton, MD, MAS (5); Helen Kim, PhD (1,2) and UCSF AHA-Bugher Center of Excellence in Hemorrhagic Stroke Research Investigators*.
Affiliations: (1) Center for Cerebrovascular Research, Dept of Anesthesia and Perioperative Care, (2) Dept of Epidemiology and Biostatistics, (3) Dept of Neurological Surgery, (4) Dept of Radiology and Biomedical Engineering, (5) Depts of Neurology & Pediatrics; University of California, San Francisco, California, USA
Correspondence concerning this article should be addressed to:
Helen Kim, PhD
University of California, San Francisco
2540 23rd St., Floor 5, Box 1363
San Francisco, CA 94143.
Email: helen.kim2@ucsf.edu
ORCID ID: 0000-0002-1937-354X
Telephone no: 415-476-2677
*UCSF AHA-Bugher Center of Excellence in Hemorrhagic Stroke Research Investigators: Adib Abla, Samyami S. Chowdhury, Daniel Cooke, Nomazulu Dlamini, Christine K. Fox, Heather J. Fullerton, Nalin Gupta, Laura Hess, Steven Hetts, Helen Kim, Yi Li, Charles McCulloch, Christine Mrakotsky, Pratik Mukherjee, Kazim Narsinh, Jeffrey Nelson, Casey Nesbit, Tomasz Nowakowski, Jarod Roland, David Saloner, Jonas Schollenberger, Alireza Sojoudi, Shantel Weinsheimer, Ethan Winkler
ACKNOWLEDGEMENTS
The authors confirm that the data supporting the findings of this study are available within the article [and/or] its supplementary materials.
This study was funded by grants to H.K. and H.J.F. from the American Heart Association and Bugher Foundation (AHA Reference # 814693, 813224) and the National Institutes of Health (R01 NS034949). The content of this article is solely the responsibility of the authors.
Conflicts of interest for authors:
H.K. is a consultant for Neurelis, Inc., Recursion Pharmaceuticals, and Ovid Therapeutics. She serves on the Data Safety and Monitoring Board for Neurelis, Imperative Care, and Route92.
Reproduced materials from third-party resources were not used in the conception of this study or manuscript.
Author Contributions:
Study concept and design: HK, SAC, SW
Patient recruitment and data collection: EW, NG, CKF, HJF
Neuroradiological review: SH, DC, YL
Statistical analysis: JN, CEM, SAC
Drafting of manuscript: SAC, SW, JN, HK
Critical revision of manuscript: All authors read and approved the final manuscript.
Copyright © belongs to author(s)
All rights reserved.
Any redistribution or reproduction of part or all of the contents in any form is prohibited other than the following:
You may print or download to a local hard disk extracts for your personal and non-commercial use only.
You may copy the content to individual third parties for their personal use, but only if you acknowledge the website as the source of the material.
You may not, except with our express written permission, distribute or commercially exploit the content. Nor may you transmit it or store it in any other website or other form of electronic retrieval system.
Abstract
Introduction: Brain arteriovenous malformations (bAVMs) are the leading cause of intracerebral hemorrhage (ICH) in children. Prior studies have implicated inflammation as a major contributor to bAVM pathogenesis and rupture, with increased inflammatory cell infiltration and interleukin protein levels reported in bAVM tissue. However, few studies have evaluated circulating inflammatory proteins as potential biomarkers for ICH in children with bAVMs.
Hypothesis: Plasma levels of inflammatory markers differ in pediatric patients with ruptured bAVMs compared to unruptured bAVMs.
Methods: This cross-sectional study included children with bAVMs (< 19 years old at presentation) evaluated at our institution between 2005 and 2022, who were enrolled in a prospective cohort study. Plasma samples (n = 52) were collected after diagnosis but before treatment and analyzed using a commercially available, comprehensive multiplex panel of 384 inflammatory markers. To control for confounders, such as the acute response to ICH, we performed segmented multivariable linear regression analysis, which allows initial protein levels to vary but assumes slopes to be constant within groups after a specified time point (e.g., 32 days). The model adjusted for age, sex, time to blood collection, and interaction of time with ICH status to identify plasma proteins associated with ICH. Exponentiated coefficients are presented and interpreted as proportional increases (PIs). We report nominally associated (p ≤ 0.05) and significantly associated proteins based on Bonferroni adjustment (p = 0.05/368 proteins = 0.00014).
Results: We analyzed blood samples collected prior to treatment in 27 unruptured bAVMs (non-ICH cases) and 25 ruptured bAVMs. The median time from study enrollment to sample collection was 126 days (IQR = 64–247) for non-ICH cases and 11 days (IQR = 4–37) for ICH cases. We observed four different patterns of association of biomarker levels with ICH status:
1) no differences between ICH and non-ICH groups over time (n=306 markers);
2) no long-term differences between ICH and non-ICH groups but initial trajectories differ after presentation (n=29);
3) long-term differences between ICH and non-ICH groups and initial trajectories differ after presentation (n=5); and
4) consistent differences between ICH and non-ICH groups over time (n=28). Thus, 33 inflammatory proteins were nominally associated (p < 0.05) with ICH either with or without initial trajectory differences (patterns 3 or 4, PI ranging from 0.71 to 4.23). Five of these 33 proteins have been associated with bAVM development and rupture in previous studies (VEGFA, MMP-10, TNF, ANGPTL2, and CSF1). No proteins were statistically significant after adjustment for multiple testing.
Conclusion: We measured a large set of inflammatory proteins in blood samples from pediatric bAVM patients and identified 33 proteins with levels nominally associated with ICH with or without initial trajectory differences, including elevated levels of VEGFA and MMP-10. Further studies are needed in larger bAVM cohorts and should include serial measurements of angiogenic or inflammatory proteins to validate the association of these potential biomarkers of ICH risk in children with bAVM.
Abstract
Introduction: Brain arteriovenous malformations (bAVMs) consist of abnormal tangles of vessels with direct connections between arteries and draining veins without an intervening capillary bed. All bAVMs have characteristic properties: one or more feeding arteries, a nidus that is the site of the arteriovenous shunt and draining venous structures.
In children, bAVMs are the leading cause of spontaneous intracranial hemorrhage (ICH) (Josephson et al., 2015). Children with a bAVM are more likely than adults (59% vs. 41%) to present with ICH despite having a similar annual rate of subsequent ICH (Choi & Mohr, 2005; Fullerton et al., 2005; Hetts et al., 2014). Pediatric bAVMs also differ from adult bAVMs with respect to the prevalence of high-risk angiographic features that are associated with ICH, e.g., exclusively deep venous drainage, a single feeding artery, smaller bAVM size, presence of venous stenosis and/or reflux, and nidal or feeding artery aneurysms (Ellis et al., 2013; Hetts et al., 2014; Oulasvirta et al., 2022). Additionally, children with bAVMs have higher recurrence rates after treatment than adults, which suggests biological differences in the dynamic nature of these lesions (Oulasvirta et al., 2022; Pezeshkpour et al., 2020).
While we do not yet fully understand the exact mechanisms contributing to ICH risk in bAVMs, several studies have pointed to the roles of abnormal angiogenesis and inflammation (Kim et al., 2011). Increased levels of inflammatory markers, such as IL-6, are found in bAVM tissue and are reportedly higher in ruptured vs. unruptured bAVM adult cases (Chen et al., 2006; Pawlikowska et al., 2004). In addition, genetic variation in IL-1β is associated with increased ICH risk (Kim et al., 2009). Matrix metalloproteinases, or MMPs, the circulating levels of which have been studied in adult patients with bAVMs, have previously been shown to be higher in patients with unruptured bAVMs compared to a control population without bAVMs; however, studies have not indicated whether MMPs are associated with ICH in bAVMs (Lattanzi et al., 2020).
While a few studies have investigated the role of inflammatory biomarkers in bAVMs, these studies have included only small numbers of children, and none have examined the association of plasma biomarkers with the risk of bAVM rupture (Fehnel et al., 2020; Ojeda-Fernandez et al., 2010; Wetzel-Strong et al., 2021). Hence, we aimed to identify circulating plasma levels of inflammatory markers associated with ICH in children with bAVMs. Inflammatory biomarkers that stratify patients into high or low-risk categories for ICH would be valuable in informing AVM treatment strategies (Germans et al., 2022; Venugopal et al., 2022).
Methods
Study Design
Patient Cohort
Patients diagnosed with a bAVM at age < 19 years were enrolled and followed prospectively at our institution between 2000 and 2022. Patients were evaluated by a multidisciplinary team that included pediatric neurosurgeons, vascular pediatric neurologists, neuro-interventional radiologists, radiation oncologists, and intensive care pediatricians. Malformations consistent with bAVMs, defined as arteriovenous shunting lesions with a nidus, were identified on brain imaging and confirmed by cerebral digital subtraction angiography. Patients were excluded from this study if they had multiple brain AVMs, spinal AVMs, dural arteriovenous fistulas, or vein of Galen malformations. The study was approved by the Institutional Review Board at the University of California, San Francisco. Parents of pediatric patients provided informed consent with patient assent when appropriate.
Study Variables
Demographic and clinical data included age, sex, race/ethnicity, and initial presenting symptoms. The clinical assessment of symptomatic presentation (including hemorrhage, seizure, headache, or focal neurological deficit) was based on formal consultation by a neurologist. The initial presentation in this study is defined as the clinical event that led to the patient presenting to the hospital with symptoms associated with bAVM (i.e., seizure, headache, or neurologic deficit) and subsequent imaging confirmation of the malformation. All patients were prospectively followed for ICH, new symptoms, and new treatments. ICH events were defined as fresh bleeding into parenchyma or CSF spaces by CT or MRI.
Digital subtraction angiography (DSA) for each case was reviewed independently by neuroradiologists and angiographic variables were recorded on predefined database forms per AVM reporting guidelines (Atkinson et al., 2001). Morphologic variables included eloquence (location in eloquent or non-eloquent brain regions), venous drainage patterns (superficial, superficial and deep, or exclusively deep), presence or absence of venous stenosis and reflux, and number of feeding arteries. Associated aneurysms were defined as the presence of shunt flow-related aneurysms (e.g., aneurysms of arteries directly supplying the AVM or subjected to increased blood flow due to the AVM) and intranidal aneurysms. The size of bAVMs was determined by radiographically measuring the maximal nidal diameter in centimeters.
Blood Sample Collection
Blood samples from bAVM cases were collected prior to any treatment (e.g., radiosurgery, embolization, or resection), typically at the time the patient was undergoing venous access for clinical purposes. EDTA plasma was isolated within one hour of collection and banked at -80°C using a standardized protocol.
Plasma Protein Analysis
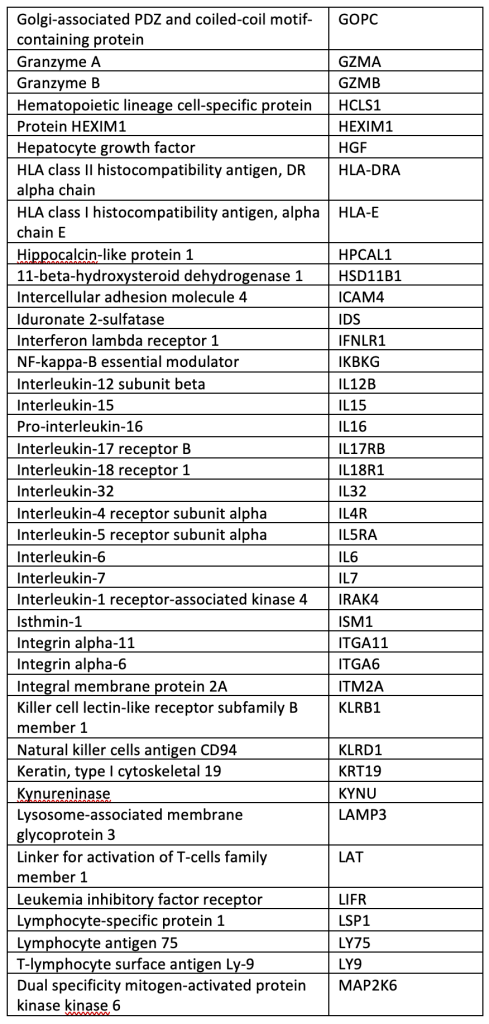
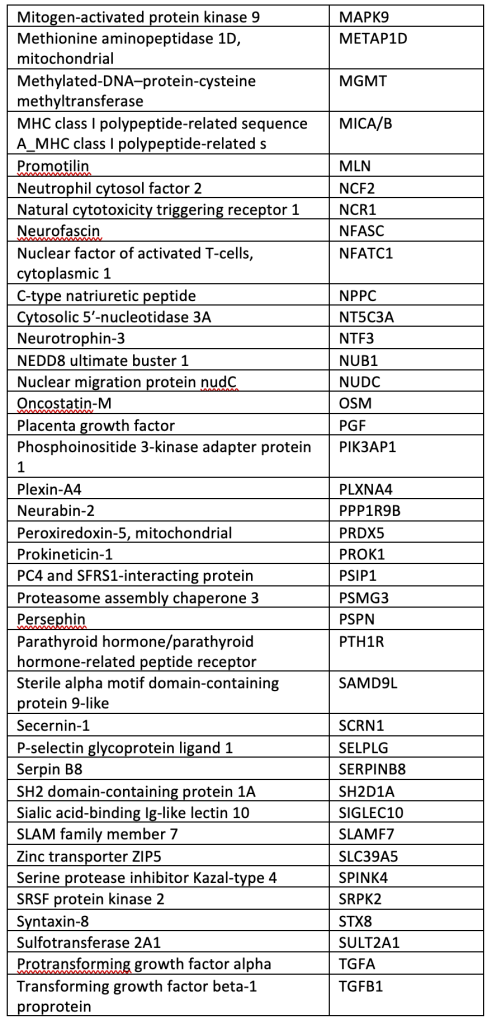
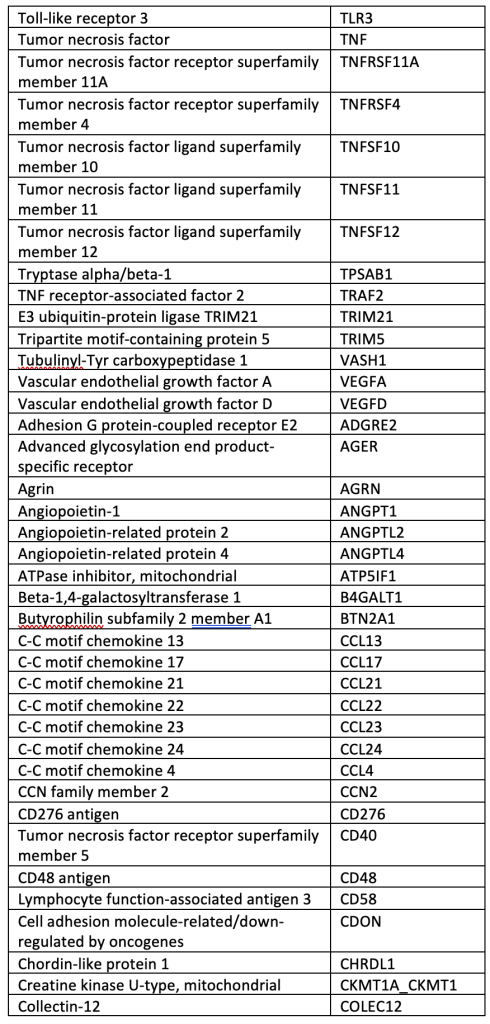
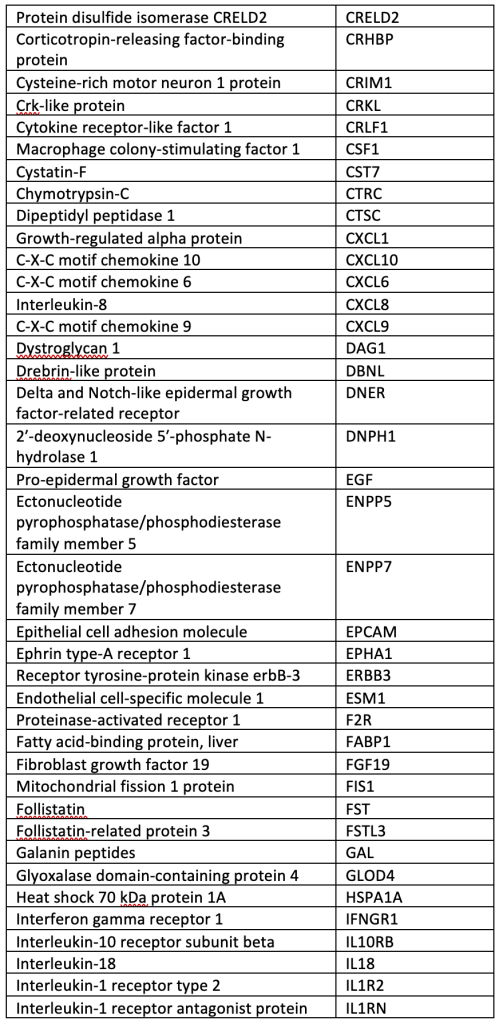
A total of 40 µL of EDTA plasma from each participant was analyzed using the OLINK® Explore 384 Inflammation panel (Supplemental Table), which contains 384 angiogenic and inflammatory proteins (OLINK, Inc., Boston, MA, USA). The panel uses a nucleic acid proximity-based assay known as the Proximity Extension Assay (http://www.olink.com/). Protein levels were quantified based on log-log standard curves; markers with >10% of values outside the range of detection were removed, and normalized protein expression (NPX) values, which are relative quantification values, were analyzed. We chose this OLINK panel because it includes many pro-inflammatory cytokines involved in bAVM pathogenesis.
Statistical Analysis
Biomarker levels vary with time or in response to acute ICH; therefore, we performed segmented multivariable linear regression to test for the association of inflammatory protein levels with ICH. Segmented linear regression fits a model that consists of connected line segments (Figures 2 and 3). This method allows for biomarker levels to vary up to a certain time point (e.g., five on a log2 scale or 32 days) and then assumes slopes are constant within groups after that time point to estimate the association between biomarker levels (outcome) and ICH (primary predictor). The model adjusts for age, sex, time from initial bAVM presentation to blood sample collection, and the interaction of time with ICH. No cases had an ICH event after the patient’s initial presentation and before blood sample collection. When the interaction term with time was not statistically significant (p < 0.05), we reran the model without the variable allowing for segmentation. Since the outcome of a given regression model was the NPX (normalized protein expression) level of the biomarker, which is an arbitrary unit on a log2 scale, exponentiated coefficients are reported as proportional increases (PI). For example, a PI = 1.3 indicates a 30% higher biomarker level in the group with ICH compared to the group without ICH, and a PI = 0.7 would indicate a 30% lower level. Plasma protein levels were considered to be significantly associated with ICH after Bonferroni correction for multiple testing of 368 proteins (p < 0.00014) and nominally associated with ICH when p < 0.05. Pairwise correlation between nominally significant ICH markers was evaluated using Pearson’s correlation coefficient and displayed using the R package pheatmap (Kolde, 2018; R Core Team, 2022). All other data analysis was conducted using Stata version 18.5 (StataCorp, 2023).
Results
Demographic and Clinical Characteristics
Among 242 children with a bAVM enrolled in the study, 157 contributed blood samples. Among these, 52 children had blood samples collected prior to any bAVM treatment and were included in the analysis. Demographic, clinical, and angiographic characteristics are summarized in Table 1. There were no differences in gender (p= 1.00), race (p = 0.42), age (p = 0.86), or ethnicity (p = 0.06) between cases with ICH (n = 25) and without ICH (n = 27) at the time of presentation. Cases without ICH initially presented with headache (n = 12, 44%), seizure (n = 10, 37%), and focal neurologic deficit (n = 8, 30%). No significant differences in angiographic characteristics (size, exclusively deep venous drainage, eloquence, degree of venous stenosis, number of feeding arteries, or associated aneurysms) were observed between groups, except for the reversal of venous outflow, which was more likely to be associated with non-ICH cases (p = 0.03).
Blood Sample Collection
The median time from study enrollment to blood sample collection was longer in the non-ICH vs. ICH groups (p = 0.02). The median time was 126 days (IQR = 64–247) for cases without ICH and 11 days (IQR = 4–37) for cases with ICH. Eight samples in ICH cases and 23 samples in non-ICH cases were collected more than 32 days after presentation.
Biomarker Analysis
Sixteen of 384 (4%) biomarkers were excluded from analyses because more than 10% of NPX values were outside the range of detection.
We identified four distinct patterns of inflammatory biomarkers in ICH cases and non-ICH cases. The first pattern was reflected in most plasma proteins (n = 306, 83.2%), which showed no difference in levels between ICH and non-ICH groups over time. For example, circulating levels of IL-1β were similar between ICH and non-ICH cases (PI = 0.90, 95% CI = 0.65-1.26, p = 0.54), as shown in Figure 1.
The second pattern, reflected in 29 (7.9%) plasma proteins, showed no long-term differences between ICH and non-ICH groups but initial trajectories differed after presentation. In other words, there was a transient elevation or depression of levels after ICH within one month of presentation but no difference between ICH and non-ICH samples collected more than one month after presentation. This was the case for IL-6 (Figure 2), where we observed an initial increase in IL-6 protein levels in ICH cases compared to non-ICH cases but no difference in IL-6 levels between groups after one month (PI = 0.91, 95% CI = 0.32-2.55, p = 0.85).
For 33 of the inflammatory proteins, there was a nominally significant difference (p < 0.05) in the marker levels between ICH and non-ICH cases (PI ranging from 0.71 to 4.23, Tables 2 and 3). For five of these proteins, a third pattern emerged in which there were long-term differences in marker levels between ICH and non-ICH groups and the initial trajectories differed after presentation (Figure 3). These five biomarkers are summarized in Table 2 and include ERBB3, TNFRSF4, LTA, TNFSF10, and LAMA4. For example, Figure 3 shows that ERBB3 levels decreased over time in non-ICH cases, whereas they transiently increased in ICH cases and then decreased but remained higher compared to non-ICH cases after one month. The fourth pattern identified 28 proteins with consistently different levels between ICH and non-ICH groups over time, as shown for LGALS9 (Figure 4, Table 3). None of the 33 biomarkers were significant after Bonferroni correction for multiple testing of 368 total proteins. The majority of these nominally associated ICH markers were positively correlated with each other (Supplemental Figure); however, most were not strongly correlated (only one pair had a correlation > 0.8).
Discussion
This study aimed to identify circulating inflammatory proteins that may be associated with ICH in children with bAVMs. We observed four different patterns of association of biomarker levels with ICH status: 1) no differences between ICH and non-ICH groups over time (n=306 markers); 2) no long-term differences between ICH and non-ICH groups but initial trajectories differ after presentation (n=29); 3) long-term differences between ICH and non-ICH groups and initial trajectories differ after presentation (n=5); and 4) consistent differences between ICH and non-ICH groups over time (n=28). Biomarkers observed in the first pattern are unlikely to be associated with ICH, as there were no differences in biomarker levels between ICH and non-ICH cases across the measured time period. Previous studies have shown that inflammatory markers associated with acute hemorrhage may show a pattern in which there is an initial elevation of the inflammatory marker level at the time of ICH or within one week of an acute ICH event, which was consistent with the second pattern of inflammatory biomarkers identified in this study (Dagonnier et al., 2021; Saand et al., 2019). However, few bAVM studies have found elevated protein levels associated with ICH that are sustained for longer than one month after the ICH event, as was shown in the third and fourth patterns of inflammatory biomarkers identified in this study. Past studies have often evaluated plasma samples collected within one week of the acute ICH, but these reports have not included an evaluation of protein levels at post-ICH time points (e.g., one week post-ICH), nor a focus on cases in which bAVMs are present (Dagonnier et al., 2021; Saand et al., 2019). Thus, the results of this study further highlight how biomarker levels can be affected by the time since ICH and how temporal evaluations of such biomarkers beyond a month from ICH could help elucidate if these biomarkers play a role beyond the acute inflammatory responses secondary to hemorrhagic events.
Our study identified 33 plasma proteins associated with ICH, either with or without differences in initial trajectories after presentation (patterns 3 and 4); however, these associations were not significant after correction for multiple testing. Five biomarkers’ patterns were consistent with the third pattern, in which there were long-term differences in marker levels between ICH and non-ICH groups, but the initial trajectory differed after presentation, perhaps reflecting an acute or secondary inflammatory response after ICH. Three of these biomarkers, TNFRSF4, LTA, and TNFSF10, have been associated with the development of intracranial aneurysms and neuroinflammation but have not been studied in bAVMs. The expression of TNFRSF4, which is thought to regulate inflammatory responses through nuclear factor kappa-beta (NFkB) pathways, was elevated in our sample and has been found to be significantly higher in intraluminal blood samples of unruptured intracranial aneurysms than in samples from control patients without aneurysms (Tutino et al., 2021). Upregulation in gene expression levels of the LTA and TNFSF10 biomarkers has also been associated with neuroinflammation through the NFkB and HMGB1 pathways, respectively, but have similarly not been investigated in studies specific to bAVM development or rupture (Cucos et al., 2022; Durocher et al., 2020).
Among the other 28 nominally significant proteins with consistent differences over time and no interaction (pattern 4), VEGFA, TNF, MMP-10, ANGPTL2, and CSF1 have been previously associated with bAVM angiogenesis and rupture. These markers may be good predictors of ICH since they were consistently increased or decreased in ICH vs. non-ICH groups. Both VEGFA and TNF were elevated in patients presenting with ICH in our sample and have previously been associated with bAVM angiogenesis, ischemic stroke, and ICH secondary to bAVM rupture (Babkina et al., 2022; Germans et al., 2022; Gong et al., 2011; Li et al., 2013). In a prior study, MMP-10 was higher in patients with unruptured bAVMs compared to a control population without bAVMs but not elevated in ICH cases secondary to bAVM rupture (Liu et al., 2022). Inflammatory markers ANGPTL2 and CSF1 may be increased in patients presenting with bAVMs because these markers are rapidly released by endothelial cells and are associated with destabilizing vascular smooth muscle cell-endothelial cell interactions during inflammatory events. However, ANGPTL2 levels have previously been shown to be reduced in patients with hereditary hemorrhagic telangiectasia (HHT) compared to control subjects without HHT, but those studies did not indicate how many of the HHT patients included had bAVMs (Ma et al., 2023; Snodgrass et al., 2021).
While other biomarkers that were elevated in patients presenting with ICH have not been studied regarding AVM pathogenesis and rupture, they have been shown to be associated with hemorrhage and ischemia secondary to neurologic events. One protein that was elevated in the samples from patients with ICH was ERBB3 (p = 0.001). ERBB1 and ERBB2 have been shown to be neuroprotective in secondary brain injury after ICH and aneurysm rupture, respectively; however, the association with ERBB3 has not been reported (Li et al., 2024; Lin et al., 2022). Levels of IFNG and GZMB are acutely increased after hemorrhagic strokes, as observed in our study, but in previous studies, this increase was usually limited to 24-96 hours after the hemorrhagic event (Germans et al., 2022; Saand et al., 2019; Stone et al., 2016; Vahidy et al., 2015). Elevation of three biomarkers, CXCL10, ANGPTL2, and IL-10 (collected between six- and 72 hours after hemorrhagic strokes), has been associated with long-term cognitive dysfunction and may be associated with ongoing brain injury, but prolonged elevation in the biomarker levels has also not been measured (Amadatsu et al., 2016; Garcia et al., 2017; Landreneau et al., 2018). Other biomarkers, including LGALS9, NCR1, and CKAP4, are often found to be elevated after ischemic strokes but have not been previously studied in hemorrhagic strokes or bAVM rupture (Braadt et al., 2023; Han et al., 2024; Wang et al., 2023). Furthermore, the inflammatory markers for ICH that were observed to be correlated (Pearson correlation coefficient > 0.5) in this bAVM cohort have not previously been studied in relation to each other. Further studies will be needed to evaluate whether coordinated levels of these proteins contribute to the pathogenesis of bAVMs, risk of bAVM ICH, or response to ICH.
Biomarkers such as GFAP, RBP-4, and S100B have previously been elevated in acute hemorrhagic stroke events and have been shown to be useful as diagnostic markers to differentiate ischemic stroke and ICH if measured within the first few hours of stroke events (Delgado et al., 2006; Katsanos et al., 2017; Llombart et al., 2016). However, these biomarkers were not included in the inflammatory panel used in this study, and thus, their association with bAVM rupture and subsequent ICH could not be determined.
While previous studies indicated an association between interleukins and bAVM pathogenesis and ICH, in this study, we found no significant difference in plasma levels of IL-1β or IL-6 in bAVM cases that present with or without ICH. IL-1β promoter polymorphisms, which have been associated with the risk of ICH during follow-up in other studies, may not necessarily be concordant with plasma levels of IL-1β, which have not been shown to be increased in patients with either ruptured or unruptured bAVMs when compared to patients diagnosed with unruptured intracranial aneurysms (Kim et al., 2009; Liu et al., 2022). Although previous studies have shown increased levels of IL-6 protein in both bAVM tissue and plasma from ruptured compared to unruptured bAVM cases, we only observed an elevation in IL-6 levels in plasma samples collected within one month of presentation in ICH cases (Chen et al., 2006; Li et al., 2013). The initial increase in IL-6 levels may be explained by the inflammatory response to an acute hemorrhage, as a transient elevation in plasma markers is associated with the inflammatory cytokine acute phase response (Chen et al., 2006; Powers et al., 2003). Our results in pediatric cases align with those of Liu et al. (2022), who found no significant difference in plasma IL-6 levels in adult bAVM patients who present with ICH when measuring these levels at least 30 days after ICH. Li et al. (2013) found an increase in plasma IL-6 levels in ICH cases; however, plasma samples in that study were collected at the time of surgical intervention to treat acute hemorrhage, and repeat samples were not collected after the intervention to evaluate how IL-6 levels may change over time.
Strengths and Limitations
Strengths of this study include the large sample size of pediatric bAVM cases with blood samples, the use of a comprehensive commercially available biomarker panel, and a careful selection of samples that were collected before bAVM treatment and over a range of time points that did not limit the analysis of biomarker levels to a week after the hemorrhage event. Limitations include the cross-sectional nature of the study. Thus, it could not be determined whether the associated plasma biomarker levels were elevated prior to bAVM rupture and could serve as predictors of ICH since all samples in the ICH group were collected after rupture. Another limitation is that our analysis assumed slopes were constant within groups after the specified time point of 32 days, which may not necessarily reflect the physiological responses to ICH events. Our sample size limited the ability to control for additional angiographic characteristics, such as venous outflow reversal and flow-related arterial aneurysms, that may confound the relationship between inflammatory levels and risk of ICH. We are also not able to comment on other important inflammatory or angiogenic markers that were not on the commercial array used.
Future Directions
To accurately stratify patients for treatment and reduce ICH risk, prospective studies in which repeat blood samples are collected from bAVM patients before and after ICH may help elucidate which biomarkers predict ICH risk. Conducting studies in which blood samples are collected at time points beyond one month after presentation would also help elucidate the association between inflammatory markers and hemorrhage in bAVM patients and how these levels may vary temporally with respect to acute and long-term inflammatory effects.
Conclusions
In this pediatric bAVM cohort, we identified 33 inflammatory biomarkers that could be further explored as markers of ICH due to the difference in biomarker levels with respect to the timing of sampling. A better understanding of the role of inflammatory markers in bAVMs could help stratify ICH risk and inform tailored management and treatment strategies.
Tables
Table 1
Clinical and imaging characteristics of 52 brain arteriovenous malformation (bAVM) patients with (+) and without (-) intracranial hemorrhage (ICH) at presentation.
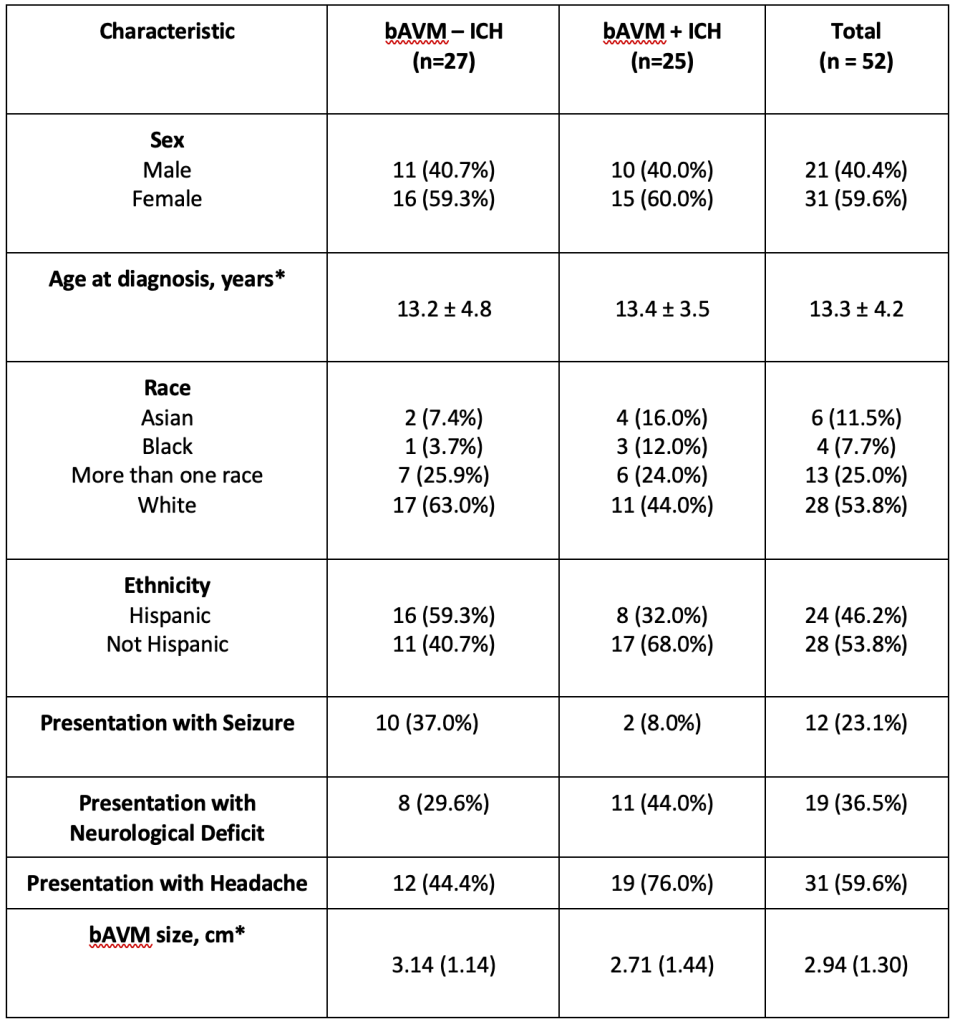
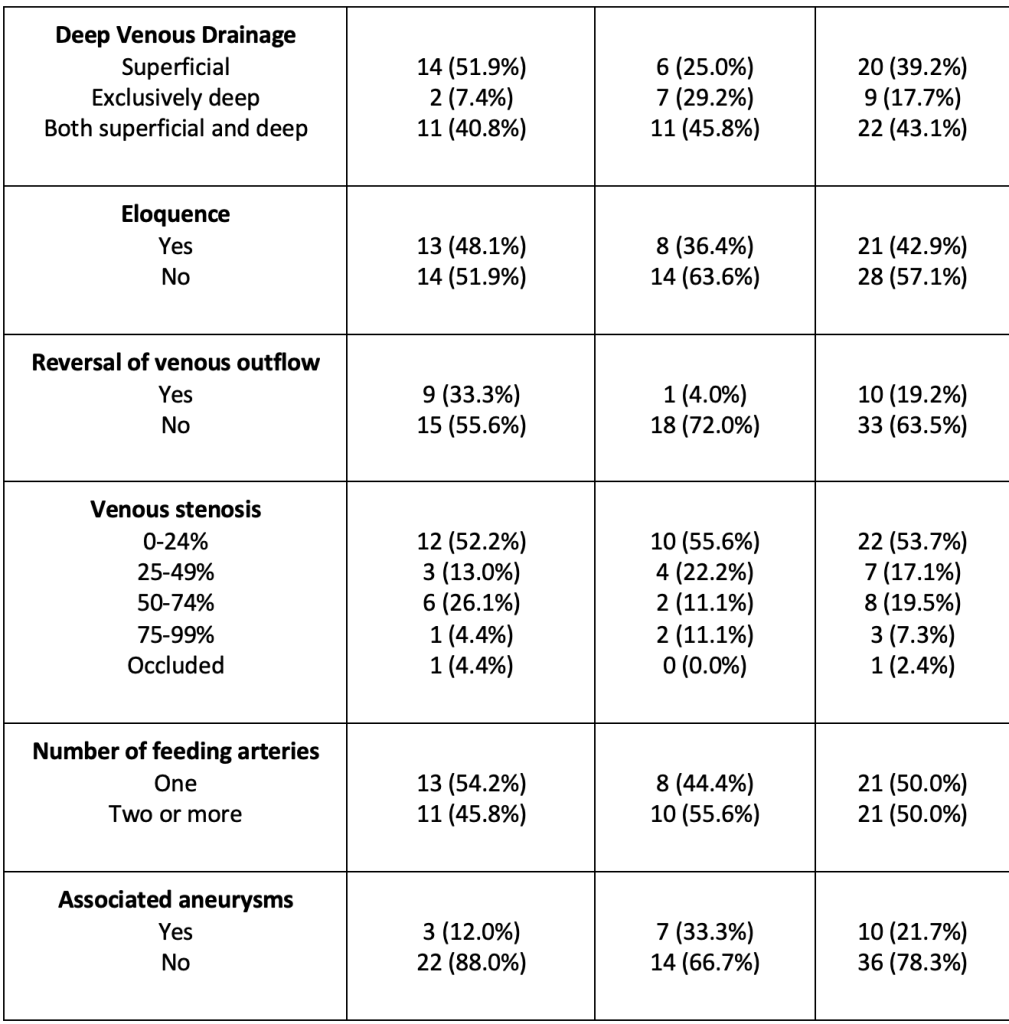
*mean +/- standard deviation
Table 2
Inflammatory markers with long-term differences (p ≤ 0.05) in levels between brain arteriovenous malformation (bAVM) intracranial hemorrhage (ICH) and non-ICH cases and with initial trajectory differences in biomarker levels measured within the first month after presentation).
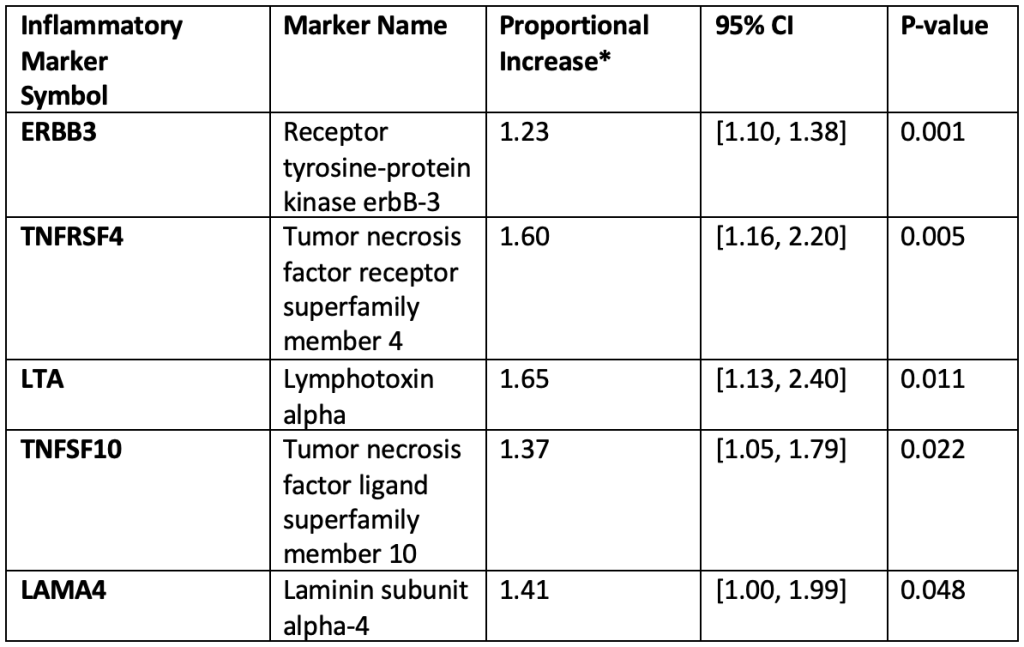
*Proportional Increase (PI) = exponentiated beta coefficient from multivariable linear regression model with segmentation.
Table 3
Inflammatory markers with consistent differences (p ≤ 0.05) in levels between brain arteriovenous malformation (bAVM) intracranial hemorrhage (ICH) and non-ICH cases over time.
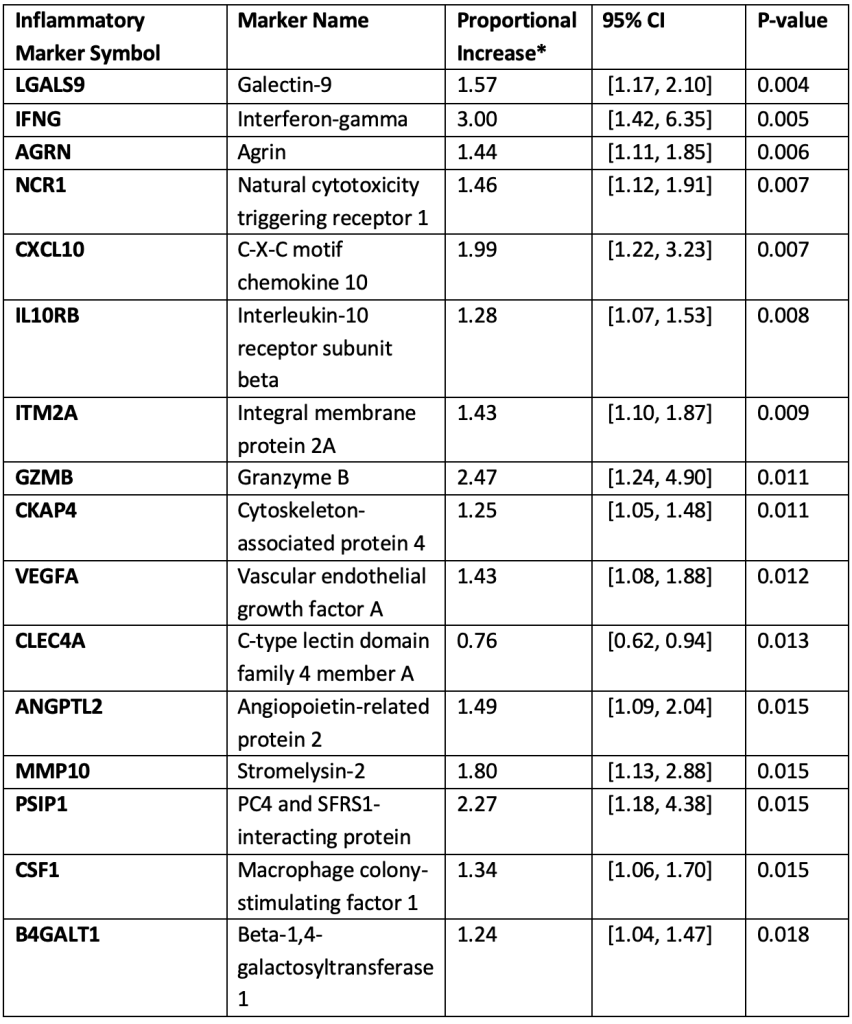
*Proportional Increase (PI) = exponentiated beta coefficient from multivariable linear regression model with segmentation.
SUPPLEMENTAL TABLE: OLINK® Explore 384 Inflammation panel.
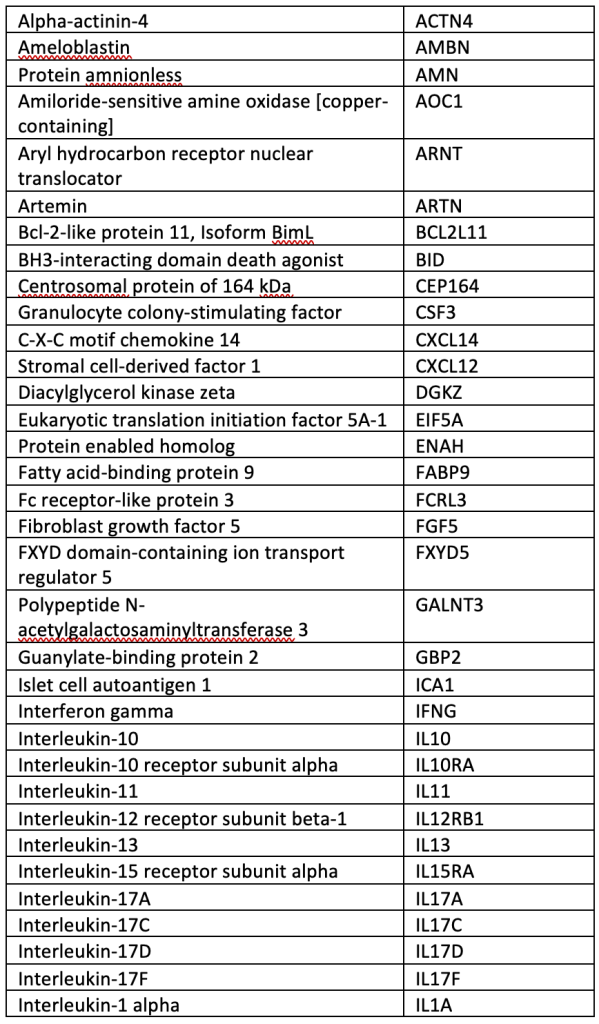
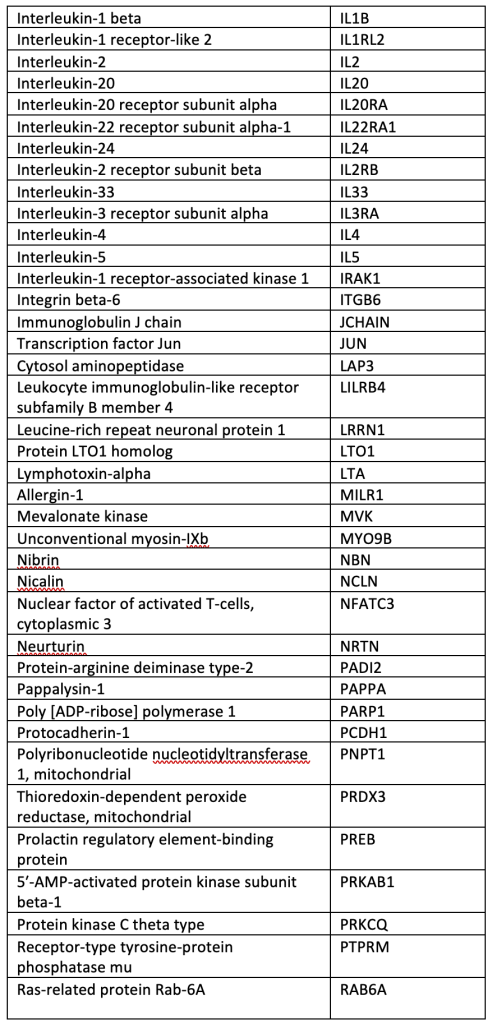
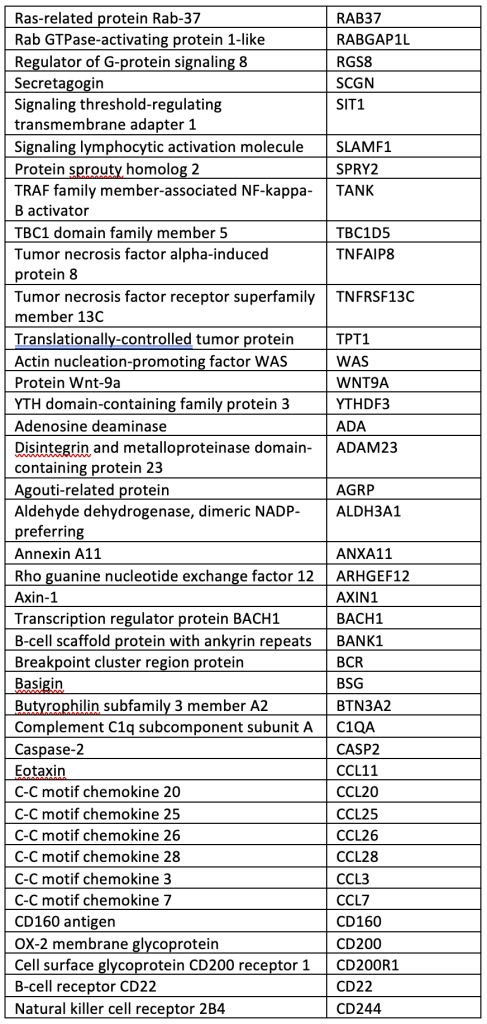
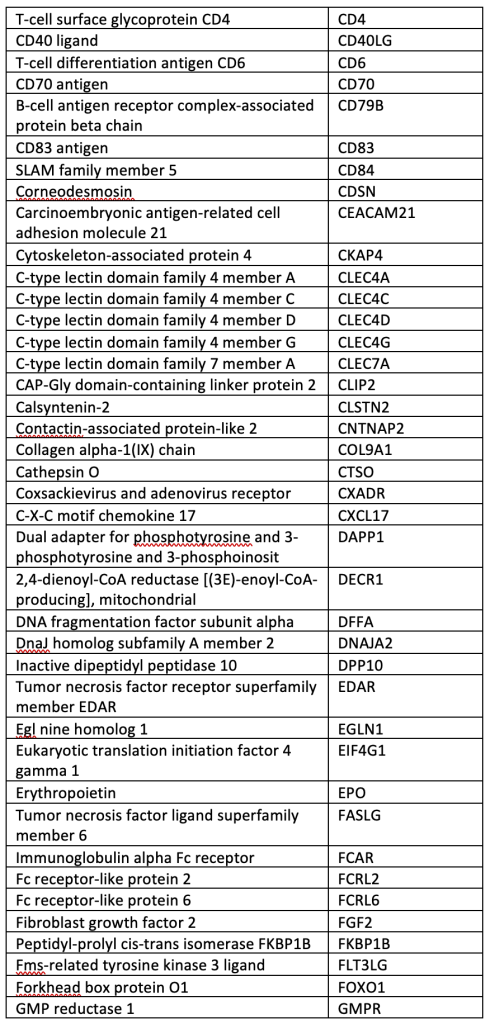
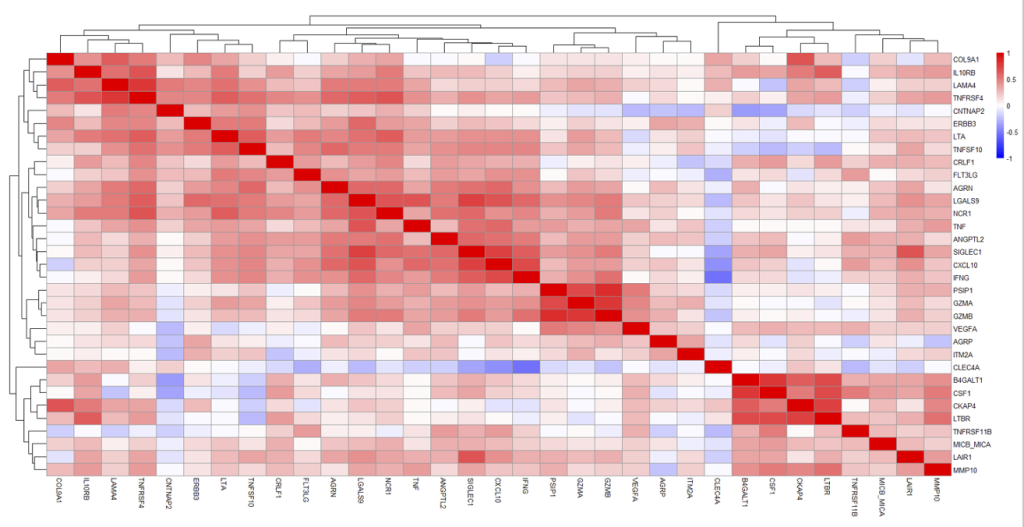
SUPPLEMENTAL FIGURE
Correlation plot (heatmap) displaying Pearson’s correlation coefficient of the 33 nominally significant ICH markers. Two distinct clusters of correlated proteins were observed in this bAVM cohort: Cluster 1 (upper left box) including COL9A1, IL10RB, LAMA4, and TNFRSF4; and Cluster 2 (lower right box) including: B4GALT1, CSF1, CKAP4, LTBR, TNFRSF11B, MICB_MICA, LAIR1, and MMP10.
References
- Amadatsu, T., Morinaga, J., Kawano, T., Terada, K., Kadomatsu, T., Miyata, K., Endo, M., Kasamo, D., Kuratsu, J., & Oike, Y. (2016). Macrophage-derived angiopoietin-like protein 2 exacerbates brain damage by accelerating acute inflammation after ischemia-reperfusion. PLOS ONE, 11(11). https://doi.org/10.1371/journal.pone.0166285
- Atkinson, R. P., Awad, I. A., Batjer, H. H., Dowd, C. F., Furlan, A., Giannotta, S. L., Gomez, C. R., Gress, D., Hademenos, G., Halbach, V., Hemphill, J. C., Higashida, R. T., Hopkins, L. N., Horowitz, M. B., Johnston, S. C., Lawton, M. W., McDermott, M. W., Malek, A. M., Mohr, J. P., Qureshi, A. I., Riina, H., Smith, W. S., Pile-Spellman, J., Spetzler R. F., Tomsick, T.A., Young, W. L. (2001). Reporting terminology for brain arteriovenous malformation clinical and radiographic features for use in clinical trials. Stroke, 32(6), 1430–1442. https://doi.org/10.1161/01.str.32.6.1430
- Babkina, A. S., Yadgarov, M. Ya., Ostrova, I. V., Zakharchenko, V. E., Kuzovlev, A. N., Grechko, A. V., Lyubomudrov, M. A., & Golubev, A. M. (2022). Serum levels of VEGF-A and its receptors in patients in different phases of hemorrhagic and ischemic strokes. Current Issues in Molecular Biology, 44(10), 4888–4901. https://doi.org/10.3390/cimb44100332
- Braadt, L., Naumann, M., Freuer, D., Schmitz, T., Linseisen, J., & Ertl, M. (2023). Novel inflammatory biomarkers associated with stroke severity: results from a cross-sectional stroke cohort study. Neurological Research and Practice, 5(1), 31.
- Choi, J. H., & Mohr, J. P. (2005). Brain arteriovenous malformations in adults. The Lancet Neurology, 4(5), 299-308.
- Chen, Y., Pawlikowska, L., Yao, J. S., Shen, F., Zhai, W., Achrol, A. S., Lawton, M. T., Kwok, P. Y., Yang, G. Y., & Young, W. L. (2006). Interleukin-6 involvement in brain arteriovenous malformations. Annals of neurology, 59(1), 72–80. https://doi.org/10.1002/ana.20697
- Cucos, C. A., Milanesi, E., Dobre, M., Musat, I. A., Manda, G., & Cuadrado, A. (2022). Altered Blood and Brain Expression of Inflammation and Redox Genes in Alzheimer’s Disease, Common to APPV717I× TAUP301L Mice and Patients. International Journal of Molecular Sciences, 23(10), 5799.
- Dagonnier, M., Donnan, G. A., Davis, S. M., Dewey, H. M., & Howells, D. W. (2021). Acute stroke biomarkers: are we there yet?. Frontiers in neurology, 12, 619721.
- Delgado, P., Alvarez Sabin, J., Santamarina, E., Molina, C. A., Quintana, M., Rosell, A., & Montaner, J. (2006). Plasma S100B level after acute spontaneous intracerebral hemorrhage. Stroke, 37(11), 2837-2839.
- Durocher, M., Knepp, B., Yee, A., Jickling, G., Rodriguez, F., Ng, K., Zhan, X., Hamade, F., Ferino, E., Amini, H., Carmona-Mora, P., Hull, H., Ander, B. P., Sharp, F. R., & Stamova, B. (2020). Molecular correlates of hemorrhage and edema volumes following human intracerebral hemorrhage implicate inflammation, autophagy, mrna splicing, and T cell receptor signaling. Translational Stroke Research, 12(5), 754–777. https://doi.org/10.1007/s12975-020-00869-y
- Ellis, M. J., Armstrong, D., Vachhrajani, S., Kulkarni, A. V., Dirks, P. B., Drake, J. M., Smith, E. R., Scott, R. M., & Orbach, D. B. (2013). Angioarchitectural features associated with hemorrhagic presentation in pediatric cerebral arteriovenous malformations. Journal of neurointerventional surgery, 5(3), 191–195. https://doi.org/10.1136/neurintsurg-2011-010198
- Fehnel, K. P., Penn, D. L., Duggins-Warf, M., Gruber, M., Pineda, S., Sesen, J., Moses-Gardner, A., Shah, N., Driscoll, J., Zurakowski, D., Orbach, D. B., & Smith, E. R. (2020). Dysregulation of the EPHRINB2−EPHB4 ratio in pediatric cerebral arteriovenous malformations is associated with endothelial cell dysfunction in vitro and functions as a novel noninvasive biomarker in patients. Experimental & Molecular Medicine, 52(4), 658–671. https://doi.org/10.1038/s12276-020-0414-0
- Fullerton, H. J., Achrol, A. S., Johnston, S. C., McCulloch, C. E., Higashida, R. T., Lawton, M. T., Sidney, S., Young, W. L., & UCSF BAVM Study Project (2005). Long-term hemorrhage risk in children versus adults with brain arteriovenous malformations. Stroke, 36(10), 2099–2104. https://doi.org/10.1161/01.STR.0000181746.77149.2b
- Garcia, J. M., Stillings, S. A., Leclerc, J. L., Phillips, H., Edwards, N. J., Robicsek, S. A., Hoh, B. L., Blackburn, S., & Doré, S. (2017). Role of interleukin-10 in acute brain injuries. Frontiers in Neurology, 8. https://doi.org/10.3389/fneur.2017.00244
- Germans, M. R., Sun, W., Sebök, M., Keller, A., & Regli, L. (2022). Molecular signature of brain arteriovenous malformation hemorrhage: a systematic review. World neurosurgery, 157, 143-151.
- Gong, Z., Qiao, N., Gu, Y., Song, J., Li, P., Qiu, H., Fan, W., Mao, Y., Chen, H., & Zhao, Y. (2011). Polymorphisms of VEGFA gene and susceptibility to hemorrhage risk of brain arteriovenous malformations in a Chinese population. Acta Pharmacologica Sinica, 32(8), 1071–1077. https://doi.org/10.1038/aps.2011.76
- Han, B., Zhou, S., Zhang, Y., Chen, S., Xi, W., Liu, C., Zhou, X., Yuan, M., Yu, X., Li, L., Wang, Y., Ren, H., Xie, J., Li, B., Ju, M., Zhou, Y., Liu, Z., Xiong, Z., Shen, L., Zhang, Y., Bai, Y., Chen, J., Jiang, W., Yao, H. (2024). Integrating spatial and single-cell transcriptomics to characterize the molecular and cellular architecture of the ischemic mouse brain. Science Translational Medicine, 16(733). https://doi.org/10.1126/scitranslmed.adg1323
- Hetts, S. W., Cooke, D. L., Nelson, J., Gupta, N., Fullerton, H., Amans, M. R., Narvid, J. A., Moftakhar, P., McSwain, H., Dowd, C. F., Higashida, R. T., Halbach, V. V., Lawton, M. T., & Kim, H. (2014). Influence of patient age on angioarchitecture of brain arteriovenous malformations. American journal of neuroradiology, 35(7), 1376–1380. https://doi.org/10.3174/ajnr.A3886
- Josephson, C. B., Rosenow, F., & Al-Shahi Salman, R. (2015). Intracranial Vascular Malformations and Epilepsy. Seminars in neurology, 35(3), 223–234. https://doi.org/10.1055/s-0035-1552621
- Katsanos, A. H., Makris, K., Stefani, D., Koniari, K., Gialouri, E., Lelekis, M., Chondrogianni, M., Zompola, C., Dardiotis, E., Rizos, I., Parissis, J., Boutati, E., Voumvourakis, K., & Tsivgoulis, G. (2017). Plasma Glial Fibrillary Acidic Protein in the Differential Diagnosis of Intracerebral Hemorrhage. Stroke, 48(9), 2586–2588. https://doi.org/10.1161/STROKEAHA.117.018409
- Kim, H., Hysi, P. G., Pawlikowska, L., Poon, A., Burchard, E. G., Zaroff, J. G., Sidney, S., Ko, N. U., Achrol, A. S., Lawton, M. T., McCulloch, C. E., Kwok, P. Y., & Young, W. L. (2009). Common variants in interleukin-1-Beta gene are associated with intracranial hemorrhage and susceptibility to brain arteriovenous malformation. Cerebrovascular diseases (Basel, Switzerland), 27(2), 176–182. https://doi.org/10.1159/000185609
- Kim, H., Su, H., Weinsheimer, S., Pawlikowska, L., & Young, W. L. (2011). Brain arteriovenous malformation pathogenesis: a response-to-injury paradigm. Acta neurochirurgica. Supplement, 111, 83–92. https://doi.org/10.1007/978-3-7091-0693-8_14
- Kolde, R. (2018). pheatmap: Pretty Heatmaps. R package version 1.0.12, https://github.com/raivokolde/pheatmap.
- Landreneau, M. J., Mullen, M. T., Messé, S. R., Cucchiara, B., Sheth, K. N., McCullough, L. D., Kasner, S. E., & Sansing, L. H. (2018). CCL2 and CXCL10 are associated with poor outcome after intracerebral hemorrhage. Annals of Clinical and Translational Neurology, 5(8), 962–970. https://doi.org/10.1002/acn3.595
- Lattanzi, S., Di Napoli, M., Ricci, S., & Divani, A. A. (2020). Matrix Metalloproteinases in Acute Intracerebral Hemorrhage. Neurotherapeutics : the journal of the American Society for Experimental NeuroTherapeutics, 17(2), 484–496. https://doi.org/10.1007/s13311-020-00839-0
- Li, X., Wang, R., Wang, X., Xue, X., Ran, D., & Wang, S. (2013). Relevance of Il-6 and MMP-9 to cerebral arteriovenous malformation and hemorrhage. Molecular Medicine Reports, 7(4), 1261–1266. https://doi.org/10.3892/mmr.2013.1332
- Li, B., Wu, J., Cao, D., Cao, C., Zhang, J., Li, X., Li, H., Shen, H., & Yu, Z. (2024). ERBB1 alleviates secondary brain injury induced by experimental intracerebral hemorrhage in rats by modulating neuronal death via plc‐γ/pkc pathway. CNS Neuroscience & Therapeutics, 30(3). https://doi.org/10.1111/cns.14679
- Lin, Y., Ma, H. Y., Wang, Y., He, J., & Liu, H. J. (2022). Identification of potential core genes for the rupture of intracranial aneurysms by a bioinformatics analysis. Frontiers in Genetics, 13, 875007.
- Liu, Y. T., Lee, C. C., Lin, C. F., Wu, H. M., Guo, W. Y., Yang, H. C., Chang, F. C., Liou, K. D., & Lin, C. J. (2022). Plasma Matrix Metalloproeteinase-9 Is Associated with Seizure and Angioarchitecture Changes in Brain Arteriovenous Malformations. Molecular neurobiology, 59(10), 5925–5934. https://doi.org/10.1007/s12035-022-02958-5
- Llombart, V., García-Berrocoso, T., Bustamante, A., Giralt, D., Rodriguez-Luna, D., Muchada, M., Penalba, A., Boada, C., Hernández-Guillamon, M., & Montaner, J. (2016). Plasmatic retinol-binding protein 4 and glial fibrillary acidic protein as biomarkers to differentiate ischemic stroke and intracerebral hemorrhage. Journal of neurochemistry, 136(2), 416–424. https://doi.org/10.1111/jnc.13419
- Ma, L., Zhu, X., Tang, C., Pan, P., Yadav, A., Liang, R., Press, K., & Su, H. (2023). CNS resident macrophages enhance dysfunctional angiogenesis and circulating monocytes infiltration in brain arteriovenous malformation. Research square, rs.3.rs-2899768. https://doi.org/10.21203/rs.3.rs-2899768/v1
- Ojeda-Fernandez, L., Barrios, L., Rodriguez-Barbero, A., Recio-Poveda, L., Bernabeu, C., & Botella, L. M. (2010). Reduced plasma levels of Ang-2 and sEng as novel biomarkers in hereditary hemorrhagic telangiectasia (HHT). Clinica Chimica Acta, 411(7-8), 494-499.
- Oulasvirta, E., Niini, T., Hafez, A., Koroknay-Pál, P., Niemelä, M., Luostarinen, T., & Laakso, A. (2022). Correlation between arteriovenous malformation nidus size and intraparenchymal hematoma volume in the event of rupture. Brain & spine, 2, 101663. https://doi.org/10.1016/j.bas.2022.101663
- Pawlikowska, L., Tran, M. N., Achrol, A. S., McCulloch, C. E., Ha, C., Lind, D. L., Hashimoto, T., Zaroff, J., Lawton, M. T., Marchuk, D. A., Kwok, P. Y., Young, W. L., & UCSF BAVM Study Project (2004). Polymorphisms in genes involved in inflammatory and angiogenic pathways and the risk of hemorrhagic presentation of brain arteriovenous malformations. Stroke, 35(10), 2294–2300. https://doi.org/10.1161/01.STR.0000141932.44613.b1
- Pezeshkpour, P., Dmytriw, A. A., Phan, K., Shroff, M. M., Dirks, P., Kulkarni, A. V., & Muthusami, P. (2020). Treatment strategies and related outcomes for brain arteriovenous malformations in children: a systematic review and meta-analysis. American Journal of Roentgenology, 215(2), 472-487.
- Powers, K. A., Woo, J., Khadaroo, R. G., Papia, G., Kapus, A., & Rotstein, O. D. (2003). Hypertonic resuscitation of hemorrhagic shock upregulates the anti-inflammatory response by alveolar macrophages. Surgery, 134(2), 312-318.
- R Core Team (2022). R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. URL https://www.R-project.org/.
- Saand, A. R., Yu, F., Chen, J., & Chou, S. H. (2019). Systemic inflammation in hemorrhagic strokes–A novel neurological sign and therapeutic target?. Journal of Cerebral Blood Flow & Metabolism, 39(6), 959-988.
- Snodgrass, R. O., Chico, T. J., & Arthur, H. M. (2021). Hereditary haemorrhagic telangiectasia, an inherited vascular disorder in need of improved evidence-based pharmaceutical interventions. Genes, 12(2), 174.
- 2023. Stata Statistical Software: Release 18. College Station, TX: StataCorp LLC.
- Stone, S. F., Armstrong, C., van Eeden, P. E., Arendts, G., Hankey, G. J., Brown, S. G., & Fatovich, D. M. (2016). Changes in differential gene expression during a fatal stroke. Journal of Clinical Neuroscience, 23, 130-134.
- Tutino, V. M., Lu, Y., Ishii, D., Poppenberg, K. E., Rajabzadeh-Oghaz, H., Siddiqui, A. H., & Hasan, D. M. (2021). Aberrant whole blood gene expression in the lumen of human intracranial aneurysms. Diagnostics, 11(8), 1442.
- Vahidy, F. S., Parsha, K. N., Rahbar, M. H., Lee, M., Bui, T.-T., Nguyen, C., Barreto, A. D., Bambhroliya, A. B., Sahota, P., Yang, B., Aronowski, J., & Savitz, S. I. (2015). Acute splenic responses in patients with ischemic stroke and intracerebral hemorrhage. Journal of Cerebral Blood Flow & Metabolism, 36(6), 1012–1021. https://doi.org/10.1177/0271678×15607880
- Venugopal, V., & Sumi, S. (2022). Molecular biomarkers and drug targets in brain arteriovenous and cavernous malformations: where are we?. Stroke, 53(1), 279-289.
- Wang, S., de Fabritus, L., Kumar, P. A., Werner, Y., Ma, M., Li, D., Siret, C., Simic, M., Li, B., Kerdiles, Y. M., Hou, L., Stumm, R., & van de Pavert, S. A. (2023). Brain Endothelial CXCL12 attracts protective natural killer cells during ischemic stroke. Journal of Neuroinflammation, 20(1). https://doi.org/10.1186/s12974-023-02689-x
- Wetzel-Strong, S. E., Weinsheimer, S., Nelson, J., Pawlikowska, L., Clark, D., Starr, M. D., … & Marchuk, D. A. (2021). Pilot investigation of circulating angiogenic and inflammatory biomarkers associated with vascular malformations. Orphanet journal of rare diseases, 16(1), 1-10.
Plasma Proteins and bAVM Hemorrhage
