Case Report
Susan De la Torre, BS1, Ian Butler, MD2, Suhas Bajgur3, Stuart Fraser, MD2
1The University of Texas McGovern Medical School; 2Department of Pediatrics, University of Texas McGovern Medical School; 3Department of Neurology, University of Texas McGovern
Corresponding Author:
Stuart Fraser, MD
Child Neurology Chief Resident
Division of Child and Adolescent Neurology
Department of Pediatrics
The University of Texas McGovern Medical School
Stuart.m.Fraser@uth.tmc.edu
Copyright © belongs to author(s)
All rights reserved.
Any redistribution or reproduction of part or all of the contents in any form is prohibited other than the following:
- you may print or download to a local hard disk extracts for your personal and non-commercial use only
- you may copy the content to individual third parties for their personal use, but only if you acknowledge the website as the source of the material
You may not, except with our express written permission, distribute or commercially exploit the content. Nor may you transmit it or store it in any other website or other form of electronic retrieval system.
Abstract
Pediatric spinal cord infarcts are rare and infrequently reported in the medical literature. As compared to brain infarcts, spinal cord infarcts are generally thought to take longer to identify. We present a case of idiopathic spinal cord infarct in an 8-year-old patient associated with posterior circulation infarcts.
Case Report
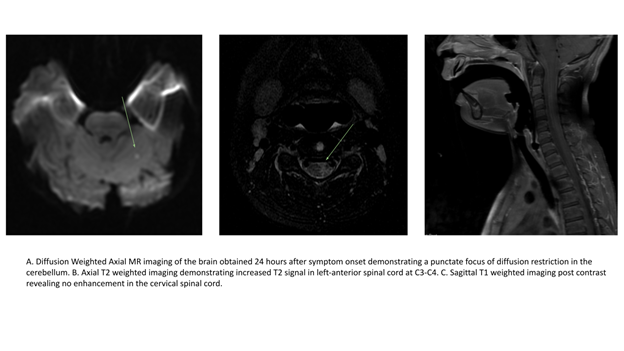
An 8-year-old male with autism presented with left arm and leg weakness, gait difficulties, and emesis on awakening. He presented to the emergency department 12 hours after symptom onset. A computed tomography (CT) brain and CT angiogram head and neck were normal. Magnetic resonance imaging (MRI) revealed abnormalities consistent with ischemic infarcts of the posterior left temporal lobe, left cerebellum, and left anterior spinal cord at the C3-C4 level. Thrombophilic, thromboembolic, and seizure evaluations were negative, and his symptoms gradually improved during hospitalization. It was considered that a vertebral artery embolism affecting the anterior spinal artery and basilar artery territories was the most likely etiology for his presentation. He was discharged on daily aspirin 81mg and continued outpatient physical rehabilitation. At follow-up two weeks later, his strength had improved, and his gait returned to baseline. Imaging two weeks later revealed normal head and neck vessels and resolution of the temporal and cerebellar punctate foci. The left spinal cord lesions were noted to be smaller, and the restricted diffusion had resolved.
Discussion
We posit that spinal cord infarction can be seen in posterior circulation stroke in children and is an important entity that pediatric neurologists should be aware of. Though Level I evidence is lacking regarding duration and treatment for idiopathic spinal cord stroke, a thorough evaluation for possible thrombotic, cardioembolic, arteriopathy, or inflammatory causes of stroke is warranted in all children with posterior circulation stroke with or without spinal cord infarction.
Case Report
Introduction
Pediatric stroke is subdivided into ischemic and hemorrhagic subtypes, and ischemic stroke can be classified as a venous or arterial ischemic infarct. Although ischemic strokes are a significant cause of morbidity and mortality in children, they are also relatively rare, occurring in an estimated 3/100,000 person-years [1]. Pediatric patients with ischemic stroke usually take longer to diagnose and treat than adults, as symptoms are often less specific, ranging from hemiparesis and hemisensory deficit to seizures and altered mental status[2]. It can be even more challenging to identify spinal cord strokes due to the wide range of possible presenting symptoms. Additionally, children may have trouble describing their deficits, and focal neurological deficits are not always present, especially in younger children and infants. Older children and adolescents with stroke commonly present with hemiplegia, language difficulties, speech difficulties, headache, and altered mental status[3]. Posterior circulation strokes, which comprise about 15% of pediatric stroke cases, can present with signs such as ataxia, vertigo, and vomiting[1]. It is believed the average delay in diagnosis of about 22 hours is related to a lack of suspicion for stroke by caregivers, low index of suspicion by emergency department personnel, high frequency of non-focal presentations, delay in obtaining imaging, low sensitivity of CT imaging for acute pediatric stroke, and a broader differential diagnosis of focal deficits in the pediatric population as compared to adults[1]. Additional challenges come from correctly identifying conditions that can also present with a focal deficit like hemiparesis, such as migraine, seizure, or post-ictal paralysis.
Comparable data for posterior circulation strokes is limited, but one retrospective study found that more than half of the cases are diagnosed more than 12 hours after symptom onset [4]. In addition to these challenges in recognizing all pediatric stroke types, posterior circulation infarcts have a pathophysiology distinct from the adult counterpart, and CT imaging may poorly visualize the posterior fossa [5].
Case Presentation
An 8-year-old Caucasian male presented to the emergency department for evaluation of new-onset left arm and leg weakness. His past medical history was significant for autism, but he was otherwise healthy. The family denied any illnesses in the months before presentation. Family history was significant for migraine headaches in many first and second-degree relatives but was otherwise nonsignificant. The patient was in his usual state of health the night before, and upon awakening the following morning, his parents noticed new onset falling to the left on ambulation and unsteady gait at approximately 8:00 AM. Four hours later, he had an episode of emesis and later in the day the parents noticed he was unable to lift his left arm while they changed his clothes at home. His parents then brought the patient to the emergency department for evaluation 8 hours after apparent symptom onset.
Upon arrival to the ED, the patient was normotensive, but his examination was notable for weakness in the left arm and leg. He was at his baseline mental status, including echolalia and frequent stereotypies. He was not able to ambulate independently. A CT scan of the head did not reveal evidence of an infarct, and computed tomography angiography (CTA) of the head and neck did not reveal any large vessel occlusion dissection. The aortic arch appeared normal in course and caliber. As his weakness was persistent, he was transferred to our children’s hospital for further evaluation.
Upon arrival, the patient had persistent left arm and leg weakness, with a Pediatric National Institute of Health Stroke Scale of 2 for left arm and leg drift. Since his examination revealed no evidence of facial weakness, MRI of the brain, cervical spine, and thoracic spine (with and without contrast), and time-of-flight angiography of the head and neck were obtained. MRI of the brain revealed four punctate abnormal foci of restricted diffusion; two in the posterior left temporal lobe and two in the left cerebellum, all of which were in the posterior circulation. MRI of the spine showed an abnormal diffusion-weighted and T2 weighted signal in the left anterior spinal cord signal for a short segment at C3-C4 (Figure 1). Inflammatory and prothrombotic laboratory workup revealed the following results as within normal limits or negative: protein C antigens, protein S antigens, Factor II mutation, Factor V PCR, homocysteine, methylmalonic acid, lupus anticoagulant, anti-cardiolipin antibodies, urine drug screen, MTHFR DNA mutation, RPR, ANA, ANCA, CRP, HIV Ag/Ab 4th Gen, TB spot, beta2-glycoprotein immunoglobulins, Coxsackie A and B antibodies, aldolase, Hemoglobin A1c, and erythrocyte sedimentation rate. Transthoracic echocardiography revealed normal cardiac anatomy and function, without visualized intra-atrial shunting of blood.
The patient’s physical examination continued to improve over the first three days of admission, including resolution of the left arm and leg drift. The patient underwent a lumbar puncture, MRI of the brain and spine, and a transesophageal echocardiogram (TEE) under general anesthesia on day 4 of admission. MRI of the cervical spine showed an interval increase in size and conspicuity of the left cord lesion at C3-C4 (Figure 2). Restricted diffusion was evident, most consistent with an evolving ischemic infarct. The punctate foci of restricted diffusion in the brain were still present, but T2 FLAIR imaging of the brain remained normal. A TEE was performed, which revealed no interatrial shunting of saline contrast at baseline, with Valsalva, and with the release of Valsalva maneuvers. The visualized aortic arch appeared normal. CSF studies revealed normal cell count, differential, and infectious and autoimmune evaluations were otherwise normal. The patient was started on aspirin 81mg daily and discharged home with plans for a short follow-up and continued outpatient rehabilitation.
During a follow-up two weeks later, his strength had returned to baseline, and his gait abnormalities resolved. The parents reported that he had been completing physical and occupational therapy twice a week and continued aspirin 81 daily. Follow-up CTA of the head and neck as well as MRI of the brain and c-spine were completed two weeks later, revealing normal head and neck vessels on CT imaging. The previously noted punctate foci of restricted diffusion in the left posterior temporal lobe and cerebellum were no longer seen on brain MRI. MRI of the cervical spine showed the anticipated interval evolution of a presumed infarct in the left paramedian cord at C3-C4 level with decreased size of the T2 signal abnormality, and resolution of the restricted diffusion. No new infarcts or abnormalities were noted. The patient continued with prophylactic medical therapy with aspirin 81mg daily.
Discussion
In this case, we demonstrate a spinal cord stroke in the anterior spinal artery distribution in association as well as small areas of diffusion restriction in the posterior circulation.
Spinal cord infarctions are rare, representing approximately ~1% of ischemic strokes. Presenting symptoms can include hemiparesis, quadriparesis, sensory deficit (including a sensory level), bowel and bladder incontinence, and autonomic dysfunction[6]. Posterior circulation strokes are more common, but still account for a minority of acute ischemic stroke cases, approximately 15-22% [4]. Data from the International Pediatric Stroke Study has demonstrated that cerebral vasculopathies are the most common risk factor of posterior circulation ischemic stroke (32%), followed by underlying chronic illnesses (30%) and acute head and neck conditions. Posterior circulation ischemic stroke is more common in male children (64%), and occurs at an average age of 7.8 years. The most common symptoms identified at presentation are limb paresis (65%), headache (54%), speech disturbance (48%), and ataxia (47%). The average pediatric National Institutes of Health stroke scale at presentation is 4. In terms of diagnosis, the median amount of time between symptom onset and confirmation is 19.8 hours. MRI is identified as the imaging modality most often required for diagnosis. Interestingly, 29% of all posterior circulation ischemic stroke patients had a normal neurologic examination at discharge. In one study, about one-third of the parents reported a full recovery at their last documented follow-up visit [7]. Although this data encompasses non-spinal cord posterior circulation ischemic stroke cases, the demographics, presentation, physical examination, and imaging findings are consistent with our case.
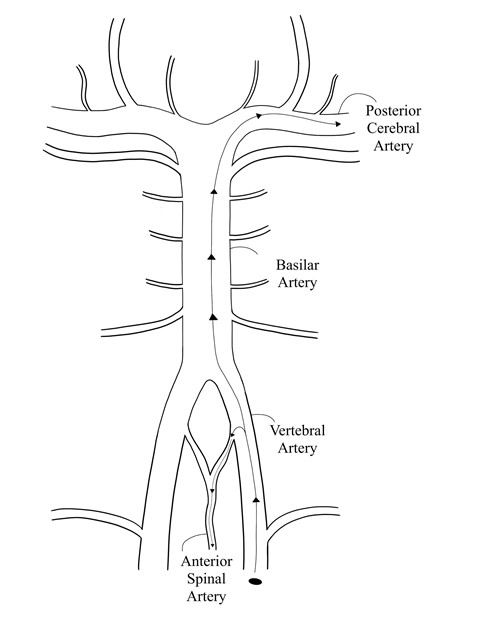
As stated above, arteriopathy is the most commonly recognized risk factor in posterior circulation pediatric acute ischemic stroke. Even so, the evaluation of pediatric posterior circulation stroke is controversial. In our case, the presence of spinal cord infarction could expand the differential diagnosis. However, we believe that, mechanistically, this case could be explained by the presence of a vertebral artery embolus. We suspect the acute ischemic changes in the left spinal cord, posterior temporal lobe, and cerebellum resulted from an embolus that ascended the left vertebral artery and migrated to the anterior spinal artery and posterior cerebral artery, respectively (Figure 3). The anterior spinal artery supplies the anterior two thirds of the cord and gives off alternating lateral branches as it descends the spinal cord. Occlusion of one of these branches can lead to hemi-cord infarction and, consequently, unilateral symptoms. Though the patient was discharged without a definite source of the presumed embolic stroke, possible causes include a small vertebral artery dissection that was not detected on imaging. Though CT angiography is often the first line patients with suspected dissection receive, it can miss small dissections. A prior review of the sensitivity of CT Angiography for vertebral artery dissection found sensitivity ranges reported as low as 51% and as high as 100% [8]. Fibrocartilaginous embolism is another cause of spinal cord stroke, and was a diagnostic consideration in this case.
Using the New England Medical Center Posterior Circulation Registry classification, we know our patient experienced proximal and distal acute ischemic strokes of the vertebrobasilar system [9]. A population-based cohort study reported that most posterior circulation arterial ischemic stroke cases were distal, while proximal involvement is the least common [4]. Interestingly, our patient’s lesions were in both the most and least common segments. Despite the presence of infarcts in three distinct locations on initial imaging, we suspect the sizeable spinal cord lesion was likely responsible for the left-sided weakness and ataxic gait. It is doubtful that the smaller temporal and cerebellar lesions would cause the severity of symptoms seen on presentation. The resolution of these lesions on follow-up imaging four weeks after discharge further supports their suspected clinical insignificance.
There has been significant research published recently regarding the evaluation of pediatric posterior circulation stroke. Notably, Dr. Fox and colleagues at the University of California San Francisco published a case series of patients with rotational arteriopathy in which several patients received curative treatment for rotational arteriopathy of the vertebral arteries and recurrent stroke [10]. Additionally, Braga and colleagues have suggested vertebral artery dissections can be safely treated with spinal fusion if rotational arteriopathy is present [11]. Unfortunately, the literature discussing how much or what type of imaging should be obtained in idiopathic posterior circulation strokes without definite arteriopathy is less well defined. In our patient, both magnetic resonance angiography and CTA did not reveal any evidence of dissection or other arteriopathy in the vertebral arteries. Though posterior circulation stroke does have a higher recurrence rate than anterior circulation stroke, the comparatively lower morbidity precludes the use of invasive neuroimaging routinely in our opinion.
Consequently, though our patient presented with an unusual stroke of an uncertain mechanism, we consider that routine MRI and thrombophilic and inflammatory evaluations are both adequate and safe for pediatric patients with a first idiopathic stroke. This is partly because surveillance MRI can detect an asymptomatic recurrent arterial ischemic stroke which would not be detected otherwise[12]. The duration of surveillance imaging is debated, but it should likely be guided by the etiology and location of the arterial ischemic stroke. For example, as stated above, a posterior arterial ischemic stroke is more likely to recur than an anterior arterial ischemic stroke. There is also evidence that primary posterior circulation arterial ischemic stroke due to a dissection makes up a large portion of recurrences [12]. Therefore, patients with an incident posterior arterial ischemic stroke may benefit from increased surveillance. The decision to obtain angiography with head tilt is controversial in pediatric posterior circulation arterial ischemic stroke. In this particular case, the patient did not receive imaging with head rotation, as we felt the risk of performing this maneuver under general anesthesia outweighed the potential benefits. As for acute treatment and management after discharge, there is a lack of relevant randomized controlled trials in the literature and no agreed-upon standard treatment [5]. Our patient was discharged on daily prophylactic aspirin 81 mg monotherapy. Other regimens include anticoagulation monotherapy and combined anticoagulation and antiplatelet therapy [12]. We trust our case will contribute to the understanding of pediatric posterior circulation strokes while also highlighting the lack of consensus regarding acute management and surveillance.
References
[1] Sejersen, Thomas., and Ching H. Wang. Acute Pediatric Neurology . 1st ed. 2014. London: Springer London, 2014. Web.
[2]Mallick AA, Ganesan V, Kirkham FJ, et al. Childhood arterial ischaemic stroke incidence, presenting features, and risk factors: a prospective population-based study. The Lancet Neurology. 2014;13(1):35-43.
Lancet Neurol. 2014;13:35–43. doi: 10.1016/S1474-4422(13)70290-4
[3] Christerson S, Strömberg B. Childhood stroke in Sweden I: incidence, symptoms, risk factors and short-term outcome: Childhood stroke. Acta Paediatrica. 2010;99(11):1641-1649.
[4] Fink, Mirjam et al. “Posterior Arterial Ischemic Stroke in Childhood: Clinical Features and Neuroimaging Characteristics.” Stroke (1970) 50.9 (2019): 2329–2335.
[5] Carey, Samantha et al. “Epidemiology, Clinical Presentation and Prognosis of Posterior Circulation Ischemic Stroke in Children.” Pediatric neurology 74 (2017): 41–50.
[6]Morshid A, Jadiry HA, Chaudhry U, Raghuram K. Pediatric spinal cord infarction following a minor trauma: a case report. Spinal Cord Ser Cases. 2020;6(1):95.
[7] Goeggel Simonetti, Rafay. “Comparative Study of Posterior and Anterior Circulation Stroke in Childhood: Results from the International Pediatric Stroke Study.” Neurology 94.4 (2020): e337–e344.
[8] Provenzale JM, Sarikaya B. Comparison of test performance characteristics of mri, mr angiography, and ct angiography in the diagnosis of carotid and vertebral artery dissection: a review of the medical literature. American Journal of Roentgenology. 2009;193(4):1167-1174.
[9] Caplan, Louis R et al. “New England Medical Center Posterior Circulation Registry.” Annals of neurology 56.3 (2004): 389–398.
[10] Fox, Christine K et al. “Single-Center Series of Boys with Recurrent Strokes and Rotational Vertebral Arteriopathy.” Neurology 95.13 (2020): e1830–e1834.
[11] Braga BP, Sillero R, Pereira RM, et al. Dynamic compression in vertebral artery dissection in children: apropos of a new protocol. Childs Nerv Syst. Published online November 6, 2020.
[12] Uohara, Michael Y et al. “Incidence of Recurrence in Posterior Circulation Childhood Arterial Ischemic Stroke.” JAMA neurology 74.3 (2017): 316–323.
Images

Spinal Cord Stroke Case Report
