Case Report
*Thao L. Nguyen, DOa,1, *Varun Kannan, MDb, Viola Caretti, MD, PhDb, Jon Cokley, PharmDb, Omar Tanweer, MDb, Daniel Davila-Williams, MDb
a. Division of Critical Care Medicine, Department of Pediatrics, Baylor College of Medicine; 6651 Main St., Houston, TX 77030, U.S.A.
b. Division of Neurology and Developmental Neurosciences, Department of Pediatrics, Baylor College of Medicine; 6621 Fannin St, Ste. 1250, Houston, TX 77030, U.S.A.
c. Department of Neurosurgery, Baylor College of Medicine; 7200 Cambridge St., Suite 9B, Houston, TX 77030, U.S.A.
1. Present Address: Division of Critical Care Medicine, Department of Pediatrics, McGovern Medical School at the University of Texas Health Science Center at Houston; 1133 John Freeman Blvd, JJL 205N, Houston, TX 77030, U.S.A.
*Both authors were equally responsible for the work described in this paper.
Abstract
While there is substantial evidence guiding management of vasospasm in adult aneurysmal subarachnoid hemorrhage, there is a lack of safety and efficacy data in the pediatric population. We describe a case report of a healthy five-month-old infant boy with severe vasospasm secondary to a traumatic pseudo-aneurysmal subarachnoid hemorrhage who underwent successful treatment using intra-arterial verapamil. The patient developed severe vasospasm as noted by a significant increase in Lindegaard ratios on transcranial doppler and confirmed by digital subtraction angiography, despite being on nimodipine for vasospasm prophylaxis. After administration of intra-arterial verapamil during angiography, the patient demonstrated objective improvement in vessel caliber with subsequent normalization of Lindegaard ratios. This patient is one of the youngest reported cases that demonstrated safe and successful use of an intra-arterial calcium-channel blocker.
Keywords: Pediatric Vasospasm, Lindegaard Ratios, Intra-arterial Verapamil, Subarachnoid Hemorrhage, Pseudoaneurysm.
Introduction
Cerebral vasospasm can cause devastating delayed cerebral ischemia (DCI) in the setting of subarachnoid hemorrhage (SAH).1 While there is substantial evidence guiding management of vasospasm in adult aneurysmal SAH, there is a lack of safety and efficacy data in the pediatric population. Here, we present a case of an infant with symptomatic cerebral vasospasm secondary to aneurysmal SAH who was successfully treated with intra-arterial (IA) verapamil.
Case Description
A previously healthy five-month-old boy presented to the emergency room with progressive lethargy after a witnessed accidental fall with cervical acceleration-deceleration injury. Initial computerized tomography (CT) of the brain (Figure 1) showed extensive subarachnoid hemorrhage in the basal cisterns and intraventricular hemorrhage with hydrocephalus, placing the patient as grade 4 with a 40% probability of symptomatic vasospasm on the Modified Fisher Scale.2 Computerized tomography angiography (CTA) identified a 6.3cm ruptured traumatic pseudoaneurysm of the right V4 vertebral artery, with extensive SAH in basal and pre-pontomedullary cisterns and intraventricular extension. We postulated that the shear injury from the acceleration-deceleration forces lead to the traumatic pseudoaneurysm and subsequent SAH.3 He was intubated due to deteriorating mental status and underwent right frontal extra-ventricular drain (EVD) placement. The pseudoaneurysm was successfully excluded with coil sacrifice of the right vertebral artery via digital subtraction angiography (DSA). He was admitted to the intensive care unit on levetiracetam for seizure prophylaxis and nimodipine (1 mg/kg every 4 hours) for vasospasm prevention. Serial transcranial doppler ultrasound (TCD) with Lindegaard ratios (LRs) and continuous quantitative electroencephalography (qEEG) were used for neuromonitoring. The LR is an index calculated by dividing the blood velocity of the middle cerebral artery (MCA) by the ipsilateral internal carotid artery (ICA).4 A ratio 3-6 suggests mild to moderate vasospasm, and ratios greater than 6 are indicative of severe vasospasm.5
Lindegaard ratios increased sharply on hospital day (HD) 8 without associated change in EEG background. Hours later he developed left flaccid hemiparesis. Magnetic resonance imaging (MRI) confirmed acute infarction mainly in the right anterior cerebral arterial (ACA) territory (Figure 2A-B). He soon developed focal right frontal electrographic status epilepticus, requiring escalation to continuous anti-seizure infusions. Examination and LRs worsened despite optimization of medical management, which included fluids to ensure euvolemia and vasoactive medications (continuous infusions of norepinephrine, epinephrine, vasopressin, and phenylephrine) to keep systolic blood pressures greater than 99%ile. On HD-10, repeat DSA confirmed severe vasospasm of the right ICA terminus (including M1 and A1 branches, Figure 1C-D), left ICA terminus, and left vertebral artery. IA verapamil was administered over 10 minutes for each infusion (5 mg [0.714 mg/kg] right ICA, 2.5 mg [0.357 mg/kg] left ICA, 2.5 mg [0.357 mg/kg] left vertebral artery) with objective improvement in vessel caliber (Figure 2C-D).
LRs decreased post-verapamil (Figure 3), and motor exam improved progressively over several days. No further seizures occurred. Nimodipine was discontinued on HD-22, and he was extubated and transferred to the acute care unit. He was discharged on HD-47 with levetiracetam, phenobarbital, and an intensive outpatient rehabilitation program. At present, he remains seizure free on levetiracetam monotherapy and is making steady motor and developmental improvement with therapies.
Discussion
Although nimodipine is used as a standardized treatment in adult SAH,1 limited pediatric data suggests modest impact on functional outcome and no clear evidence for DCI prevention.6-7 In our case, serial LR measurements suggested the presence of vasospasm before any changes were appreciated in qEEG alpha-delta ratio trending. Quantitative EEG patterns including alpha-delta ratio trends have been validated as markers of cerebral perfusion in pediatric severe TBI,8 but data in pediatric SAH and DCI is lacking. Adult studies have shown reliable use of qEEG patterns such as alpha power, relative alpha variability, and alpha/delta ratio to detect DCI.9 In addition, there is growing interest to use EEG to detect cortical spreading depolarization, a wave of depolarization that happens after neuronal injury causing increased metabolic demand, DCI, and a vicious of cycle of further neuronal injury and death.9 As such, the use of qEEG in pediatric SAH and DCI has great potential for further clinical application and research.
Angiographic intervention for vasospasm refractory to medical management has demonstrated efficacy in adult SAH,1 but data in the pediatric population is limited to individual cases. Nahed et al reported administration of IA-verapamil in a 22-month-old child with posterior circulation vasospasm and DCI resulting in improvement in qualitative vessel diameter and motor weakness.10 Conversely, Snelling et al attempted IA-verapamil in an 8-month-old child with anterior circulation vasospasm with no response.11 However, they did report marked improvement in vessel caliber and TCD velocities with mechanical angioplasty.11 In both cases, and in our patient, IA-verapamil and angiography were well-tolerated. The overall benefit of angiography reducing delayed cerebral ischemia and long-term neurological deficits outweighs the risk of angiography, with complications arising in only 6.5% of cases in a study by Lauzier et al.12 The majority had minor complications (6.0%) that included headaches, nausea/vomiting, and hypotension. The other 0.5% (3 cases) had major complications, which included 1 case of thalamic infarct and 2 cases of anaphylaxis.12
In summary, we describe an infant with aneurysmal-SAH and DCI who demonstrated robust sonographic and clinical improvement following IA-verapamil treatment. This case also supports the use of TCD and LR measurements as a vasospasm screening tool and as an objective measure of IA-verapamil treatment response in pediatric SAH. Additional studies are needed to further elucidate optimal management of pediatric vasospasm.
References
Etminan N, Macdonald RL. Management of aneurysmal subarachnoid hemorrhage. Handb Clin Neurol. 2017;140:195-228. doi: 10.1016/B978-0-444-63600-3.00012-X. PMID: 28187800.
Claassen J, Bernardini GL, Kreiter K, Bates J, Du YE, Copeland D, Connolly ES, Mayer SA. Effect of cisternal and ventricular blood on risk of delayed cerebral ischemia after subarachnoid hemorrhage: the Fisher scale revisited. Stroke. 2001 Sep;32(9):2012-20. doi: 10.1161/hs0901.095677. PMID: 11546890.
Miley JT, Rodriguez GJ, Qureshi AI. Traumatic Intracranial Aneurysm Formation following Closed Head Injury. J Vasc Interv Neurol. 2008 Jul;1(3):79-82. PMID: 22518228; PMCID: PMC3317295.
Lindegaard KF, Nornes H, Bakke SJ, Sorteberg W, Nakstad P. Cerebral vasospasm diagnosis by means of angiography and blood velocity measurements. Acta Neurochir (Wien). 1989;100(1-2):12-24. doi: 10.1007/BF01405268. PMID: 2683600.
Kirsch JD, Mathur M, Johnson MH, Gowthaman G, Scoutt LM. Advances in transcranial Doppler US: imaging ahead. Radiographics. 2013 Jan-Feb;33(1):E1-E14. doi: 10.1148/rg.331125071. PMID: 23322845.
Heffren J, McIntosh AM, Reiter PD. Nimodipine for the prevention of cerebral vasospasm after subarachnoid hemorrhage in 12 children. Pediatr Neurol. 2015 Mar;52(3):356-60. doi: 10.1016/j.pediatrneurol.2014.11.003. Epub 2014 Nov 11. PMID: 25585913.
McGowan B, Khaira G, Coghlan MA, Shaibani A, Alden TD, Pardo AC. Use of Nimodipine in a Neonate With Cerebral Vasospasm With Delayed Ischemia From Subarachnoid Hemorrhage in the Posterior Fossa. Pediatr Neurol. 2020 Oct;111:44-45. doi: 10.1016/j.pediatrneurol.2020.06.018. Epub 2020 Jul 3. PMID: 32951659.
Appavu B, Foldes S, Temkit M, Jacobson A, Burrows BT, Brown D, Boerwinkle V, Marku I, Adelson PD. Intracranial Electroencephalography in Pediatric Severe Traumatic Brain Injury. Pediatr Crit Care Med. 2020 Mar;21(3):240-247. doi: 10.1097/PCC.0000000000002136. PMID: 31569184.
Baang HY, Chen HY, Herman AL, Gilmore EJ, Hirsch LJ, Sheth KN, Petersen NH, Zafar SF, Rosenthal ES, Westover MB, Kim JA. The Utility of Quantitative EEG in Detecting Delayed Cerebral Ischemia After Aneurysmal Subarachnoid Hemorrhage. J Clin Neurophysiol. 2022 Mar 1;39(3):207-215. doi: 10.1097/WNP.0000000000000754. PMID: 34510093; PMCID: PMC8901442.
Nahed BV, Ferreira M, Naunheim MR, Kahle KT, Proctor MR, Smith ER. Intracranial vasospasm with subsequent stroke after traumatic subarachnoid hemorrhage in a 22-month-old child. J Neurosurg Pediatr. 2009 Apr;3(4):311-5. doi: 10.3171/2008.12.PEDS08206. PMID: 19338410.
Snelling BM, Sur S, Shah SS, Peterson EC. Treatment of Cerebral Vasospasm in an Infant Using a Modified Dotter Technique. J Cerebrovasc Endovasc Neurosurg. 2017 Mar;19(1):48-51. doi: 10.7461/jcen.2017.19.1.48. Epub 2017 Mar 31. PMID: 28503488; PMCID: PMC5426190.
Lauzier DC, Osbun JW, Chatterjee AR, Moran CJ, Kansagra AP. Safety of pediatric cerebral angiography. J Neurosurg Pediatr. 2021 Nov 5;29(2):192-199. doi: 10.3171/2021.8.PEDS21301. PMID: 34740194.
Figures’ Caption
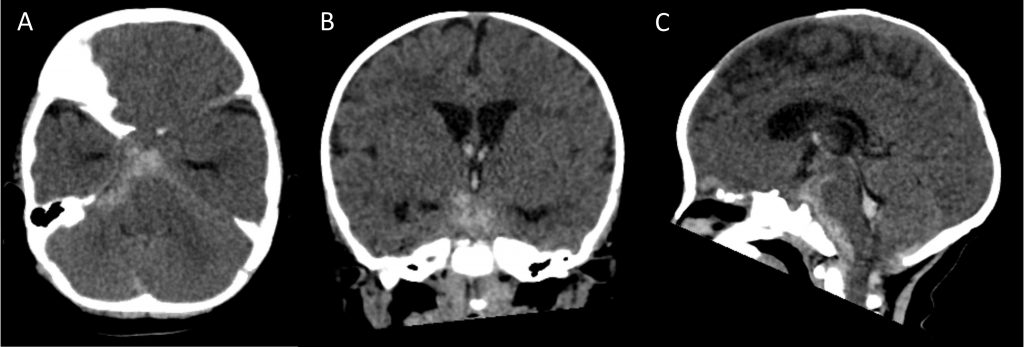
Figure 1: Initial computerized-tomography (CT) scan of the brain showing acute subarachnoid hemorrhage centered in the basal cisterns and intraventricular hemorrhage with mild hydrocephalus. This is Grade 4 on the Modified Fisher Scale for subarachnoid hemorrhage. Axial (A), coronal (B), and sagittal (C) views.
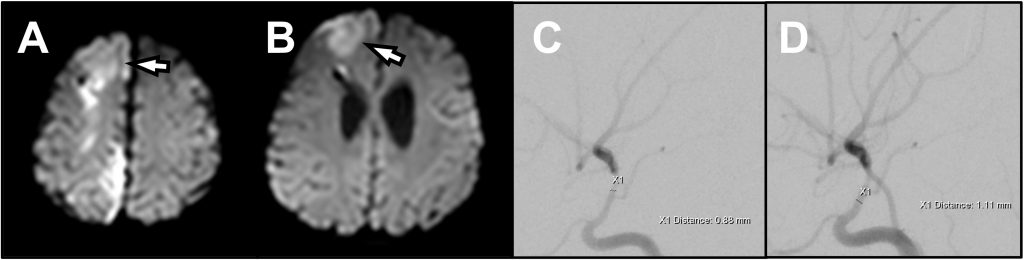
Figure 2: (A, B) Axial diffusion-weighted magnetic resonance imaging showing acute cytotoxic edema in right parasagittal frontal and parietal lobes in an anterior cerebral artery distribution consistent with vasospasm-induced ischemia. (C) Pre- and (D) Post-IA verapamil angiography of the right internal carotid artery, demonstrating improvement in vessel caliber and distal flow after treatment.
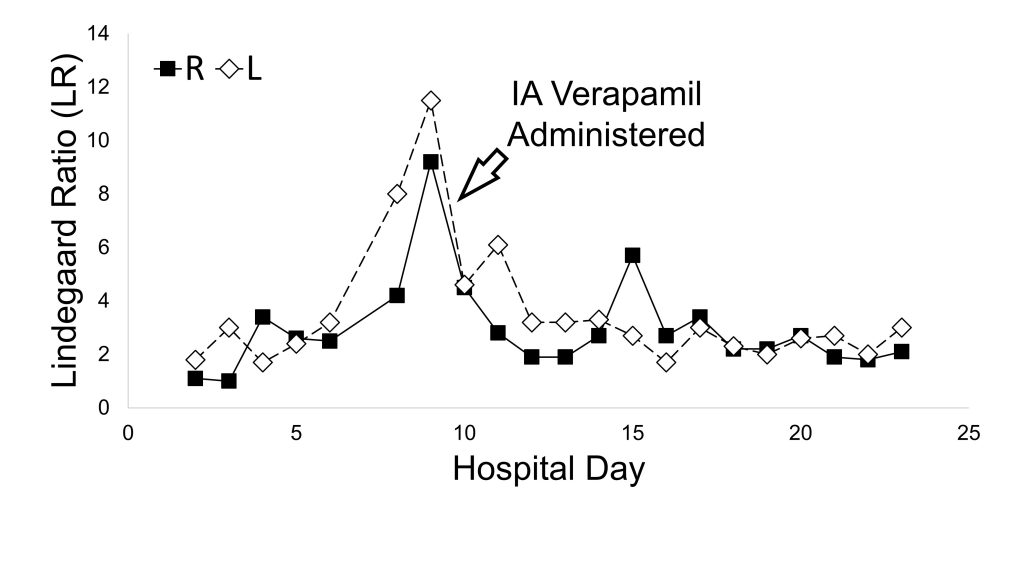
Figure 3: Lindegaard ratios calculated from serial transcranial Doppler ultrasound measurements, with arrow marking the date of intra-arterial calcium channel blocker treatment.
Verapamil for pediatric vasospasm
