Review
A review of current practices, outcomes, and future directions.
Janette A Mailo1, Ratika Srivastava2, Francois D Jacob1, Adam Kirton3
- Division of Pediatric Neurology, Department of Pediatrics, University of Alberta. AB, Canada
- Calgary Pediatric Stroke Program, University of Calgary, Calgary, AB, Canada.
- Department of Pediatrics, University of Calgary, Calgary, AB, Canada; Department of Clinical Neuroscience, University of Calgary, Calgary, AB, Canada; Department of Radiology, Faculty of Medicine, University of Calgary, Calgary, AB, Canada; Alberta Children’s Hospital Research Institute, Calgary, AB, Canada; Hotchkiss Brain Institute, Calgary, AB, Canada; Calgary Pediatric Stroke Program, University of Calgary, Calgary, AB, Canada.
Correspondence: jmailo@ualberta.ca
Copyright © belongs to author(s)
All rights reserved.
Any redistribution or reproduction of part or all of the contents in any form is prohibited other than the following:
- you may print or download to a local hard disk extracts for your personal and non-commercial use only
- you may copy the content to individual third parties for their personal use, but only if you acknowledge the website as the source of the material
You may not, except with our express written permission, distribute or commercially exploit the content. Nor may you transmit it or store it in any other website or other form of electronic retrieval system.
Abstract
Perinatal strokes are disorders of cerebrovascular origin that occur in the developing brain between 20 weeks of gestation and 28 days of postnatal life. This review aims to describe specific perinatal stroke diseases, possible risk factors, clinical presentations and management, followed by a review of outcomes and strategies focused on optimizing quality of life for affected children and their families. Perinatal stroke can present acutely and should be considered for any newborn with unexplained encephalopathy. Clinical presentation of perinatal stroke can also occur later in childhood. In either case, subsequent neuroimaging can be used to precisely identify a specific perinatal stroke syndrome. In recent years, we have seen significant advances not only in better understanding of mechanisms of early cerebrovascular brain injuries but also of the impact of rehabilitation on recovery. Many survivors of perinatal stroke will have long-term morbidity, which can include cerebral palsy, epilepsy, learning problems, cognitive and behavioral challenges, and visual deficits. With limited preventative options, neurorehabilitation will continue being the main focus for improved outcomes. Equally important is the significant psychosocial impact of perinatal stroke on the entire family. Online resources and support systems are increasingly available through national and international pediatric stroke organizations, with the goal to ensure access for all affected and interested patients and families.
Introduction
Perinatal stroke is a cerebral injury of vascular origin occurring between 20 weeks of gestation and 28 days of postnatal life (Raju, 2007). Diagnosis is most often made after an acute presentation in the neonatal period or may be retrospective, when clinical presentation occurs later in childhood. Acute perinatal stroke should be included on the differential diagnosis in any newborn with unexplained encephalopathy, seizures, lethargy, hypotonia, feeding difficulties, apnea, or focal neurological deficits. Diagnosis is confirmed by neuroimaging which can precisely identify one of multiple specific perinatal stroke diseases, providing valuable information regarding potential mechanisms, treatment, and prognosis. Perinatal stroke diseases are defined by clinical and radiographic features such as timing of presentation, arterial versus venous territory, and ischemic versus hemorrhagic injury. Though little is understood about the pathophysiology of many perinatal strokes, disease-specific treatments and neuroprotective strategies may improve outcome. Most survivors will suffer lifelong morbidity including cerebral palsy in the majority and additional complications in 25% or more including epilepsy, language and learning delays, and behavioral and mental health problems. The outcomes of perinatal stroke can have a major impact on the entire family.
This review will focus on some of the most pressing issues in perinatal stroke in the fetus and term newborn. Stroke in the preterm infant is reviewed elsewhere and global hypoxic-ischemic brain injury does not meet the definition for perinatal stroke. We will define each specific perinatal stroke disease and discuss their presentation, diagnosis, possible risk factors and management followed by a review of possible outcomes and strategies to optimize participation and quality of life for affected children and families.
SYNDROMIC CLASSIFICATION OF PERINATAL STROKE DISEASES
Acute symptomatic perinatal strokes are identified in newborns who typically present within the first days after birth with focal seizures or encephalopathy. The three acute perinatal stroke diseases are neonatal arterial ischemic stroke (NAIS), neonatal hemorrhagic stroke (NHS), and neonatal cerebral sinovenous thrombosis (CSVT). It’s important to mention that the terms perinatal arterial ischemic stroke (PAIS) or perinatal ischemic stroke (PIS) are also used (Martinez-Biarge, 2019). Understanding the difference between PAIS/PIS and NAIS is worth briefly discussing since the terms should not be used interchangeably. While PAIS/PIS include strokes occurring between 20 weeks of gestation, and the first 7 days after birth, NAIS covers the time from birth until the 28th day of postnatal life. The term PAIS/PIS then includes neonates with strokes occurring prenatally, many of whom would be asymptomatic at birth. However, neonates with NAIS will be symptomatic during the neonatal period and receive the diagnosis in the first four weeks of life. It’s important to also keep in mind that strokes after the perinatal period, the first 7 days of life, will likely have different etiologies than true perinatal strokes (Martinez-Biarge, 2014).
The remaining perinatal stroke types present in a delayed manner, often first apparent at 4-6 months of age when early hand preference or other motor asymmetry is appreciated as hemiparetic cerebral palsy. Subsequent neuroimaging then confirms a remote, chronic infarct and presumed perinatal ischemic stroke which may be arterial (APPIS) or a periventricular venous infarction (PVI) (Dunbar, 2018).
NEONATAL ARTERIAL ISCHEMIC STROKE
Neonatal arterial ischemic stroke (NAIS) refers to arterial ischemic strokes that present in term neonates within the first 28 days of life. NAIS is not rare; its estimated incidence is >1:3000 live births (Dunbar, 2020). This exceeds the incidence of childhood stroke by approximately 10-fold, bringing it up to second only to the incidence of ischemic stroke in older adults. (Armstrong-Wells 2009, Govaert P). NAIS is the most common type of perinatal stroke and represents approximately 70-80% of all symptomatic perinatal stroke cases (Lehman 2017, Ferriero 2019).
PATHOPHYSIOLOGY AND RISK FACTORS
Pathogenesis of NAIS is not well understood. Pathophysiological mechanisms of perinatal arterial ischemic stroke have been considered under large categories including emboli of cardiac and extra-cardiac origin, thrombosis due to disturbed homeostasis, and arteriopathies. NAIS uniquely involves not just the neonate, as maternal, obstetrical, placental and fetal factors can also be taken into consideration. A number of modestly powered, controlled studies have suggested possible clinical risk factors (Dunbar, 2019), but definite causative mechanisms remain difficult to establish in many cases and multiple mechanisms may coexist in an individual neonate.
Current evidence suggests that placental embolism is the most common cause of NAIS. A long list of placental pathologies includes infectious (chorioamnionitis), inflammatory, and thrombotic disorders of the fetal side of the placental circulation (Lee 2005, Elbers 2011, Sorg 2020, Roy 2020). Biological plausibility is further supported by the common occurrence of bilateral lesions, suggesting a proximal embolic source while cardiac examination is normal, and the extremely low risk of recurrence (Mineyko, 2011). Though often difficult to obtain, the placenta should be sent for pathological examination whenever possible as it may provide evidence of a definitive cause.
In only a minority of NAIS cases, a clear causative factor can be identified. Complex congenital heart disease and bacterial meningitis are well established causes of NAIS that can usually be quickly and definitively diagnosed clinically with specific investigations (echocardiography, lumbar puncture), prompting the initiation of specific treatments. Prothrombotic factors are also commonly considered due to the evolutionarily adaptive mechanisms in coagulation systems that occur during the birth process and in the early postnatal period (Nelson 2004, Wu 2005, Ferriero 2019). Prothrombotic disorders may contribute to increased risk of perinatal ischemic stroke but the evidence for direct association is relatively weak (Lehman, 2017). Known prothrombotic conditions have been investigated in neonatal arterial ischemic stroke, including decreased levels of protein C, protein S and antithrombin, increased levels of factors VIII, IX, XI, fibrinogen, homocysteine anticardiolipin antibodies or lipoprotein A, and presence of lupus anticoagulant, factor V Leiden and prothrombin G20210A gene mutation. A prospective population-based case-controlled study encompassing 212 neonates with perinatal stroke and 77 controls showed that thrombophilia in children with perinatal stroke is rare, with rates similar to the normal population (Curtis, 2017). The authors did not exclude the possibility of disordered coagulations at the time of the stroke but suggested that thrombophilia evaluation later in childhood is not indicated. A retrospective study of 215 infants with perinatal stroke concluded that even if present, neither thrombophilia, arteriopathy, nor cardioembolic risk factors were predictive of recurrent perinatal stroke (Lehman 2017). The extremely low recurrence rate after perinatal arterial ischemic stroke supports the role of mechanisms present only prenatally or perinatally (Kirton 2015, Lehman 2017).
Many additional clinical and laboratory factors have been examined for possible associations with NAIS, but the consistency of results and strength of correlations have been weak. Maternal factors suggested but not well proven may include maternal autoimmune and prothrombotic abnormalities including antiphospholipid syndrome (Akanli, 1998), history of infertility, preeclampsia, prolonged rupture of membranes (Lee, 2005), diabetes, intrapartum fever, infection (Martinez-Biarge, 2016), smoking during pregnancy (Darmency-Stamboul, 2012), and cocaine abuse during pregnancy (Chasnoff, 1986). It should be noted that some of these suggestions are based on case reports or small cases series while other common conditions such as diabetes have shown minimal associations in only some studies. Fetal factors may include intrauterine growth restriction (Lee 2005, Sorg 2020), preterm birth (Sorg 2020) and twin-twin transfusion syndrome (Cheong, 2009). Additional neonatal factors may include sepsis, polycythemia, disseminated intravascular coagulation, and rare arteriopathies or inborn errors of metabolism (Gunther 2000, Lequin 2004, Elbers 2011, Harteman, 2012). A recent controlled study demonstrated unique inflammatory profiles in babies with NAIS as compared to fetal strokes and controls (Mineyko, 2020). Uncommon but increasingly recognized genetic conditions have been associated with some forms of perinatal stroke, such as COL4A1 mutations (Takenouchi, 2015). There is no evidence that mechanical factors or “birth trauma” can cause NAIS. Many non-specific factors surrounding delivery including fetal heart rate abnormalities, presence of meconium, assisted delivery, low APGAR scores, and others have been associated with NAIS. These lack a biologically plausible mechanism and instead may represent associations with other more likely causative mechanisms such as infection, systemic disease, or placental pathology.
DIAGNOSIS
NAIS is a clinical and radiographic diagnosis, often made after seizures or encephalopathy leads to neuroimaging that reveals acute ischemic stroke in an arterial territory. Seizures are the presenting symptom in the majority, and onset of focal motor seizures 12 hours or more after birth have been shown to be independent predictors of stroke (Rafay, 2009). Focal deficits such as hemiparesis may be observed in neonates but are uncommon and not reliable for excluding stroke. All acute forms of perinatal stroke should be considered in the differential diagnosis of the newborn with unexplained seizures or encephalopathy, and prompt imaging sought to confirm or exclude.
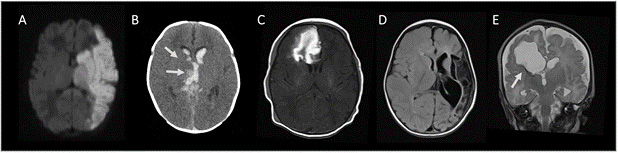
Diagnosis of a perinatal stroke is confirmed by neuroimaging and MRI is the modality of choice. Consensus based imaging protocols have been developed to facilitate accurate and timely diagnosis of neonates with suspected stroke (Lee, 2017). Magnetic resonance imaging (MRI) is ideally suited with basic sequences providing high sensitivity and specificity to confirm stroke type, number, and location without radiation exposure or need for anesthesia. Core recommended sequences include diffusion-weighted imaging and apparent diffusion coefficient mapping to diagnose acute ischemia. Large infarcts within the middle cerebral artery (MCA) territory are most common (Figure 1a) but DWI can detect very small infarcts in any location. Additional useful sequences include gradient-recalled echo or susceptibility-weighted imaging to detect hemorrhage and standard T1- and T2-weighted images (Lee, 2017). The full extent of the infarction on DWI may evolve during the first week but repeat MRI is seldom indicated.
Magnetic resonance angiography (MRA) is helpful to assess for arterial abnormalities. The arterial occlusion may be visible on MRA though spontaneous recanalization has often already occurred. MRA of the head and neck can also detect congenital arteriopathies which are rare, and screen for arterial injury (dissection), though definitive evidence of this mechanism remains unreported despite over 1000 neuroimaging cases of NAIS in the literature.
MRI is not always feasible, particularly in critically ill neonates, due to its need for transportation to the radiology suite and longer scanning time. Cranial ultrasonography can be easily done at bedside and is helpful to screen for large hemorrhages or other lesions when a stroke diagnosis is being considered but sensitivity for NAIS is modest (Golomb, 2003). Head CT can serve a similar purpose when MRI is not readily available and acute intracranial pathology needs to be excluded.
MANAGEMENT
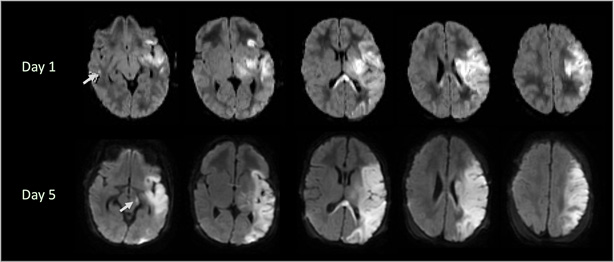
Once stroke in a newborn is diagnosed, focus should be directed at neuroprotection to minimize brain injury and the identification of any treatable underlying conditions. Neuroprotective strategies are based mostly on preclinical evidence and theory that suggest modifiable factors can influence the progression of brain cell death over days and even weeks in the newborn (Ferriero, 2019). While serial imaging is not usually required, it can demonstrate the progression of stroke injury over time (see Figure 2).
Supportive measures include seizure control, adequate oxygenation, correction of dehydration and anemia, and correction of metabolic disturbances including acidosis, hypoglycemia, and electrolyte disorders (Ferreiro, 2019). While difficult to study, seizures have been independently associated with worse outcome in animal and human studies of neonatal brain injury and should therefore be carefully monitored and promptly treated (Kharoshankaya, 2016). Clinical identification of seizures in neonates is difficult and continuous neuromonitoring using video electroencephalography (EEG) or other cerebral function monitoring (Shellhaas, 2011) may be advantageous. Diagnosing seizures in neonates with acute stroke on a clinical basis only increases the risk of both underdiagnosis and overdiagnosis. There is no stroke-specific evidence regarding choice of antiseizure medication but recent trials in related HIE populations suggest phenobarbital may be superior to levetiracetam (Sharpe, 2020). The natural history of acute stroke-related seizures in the neonate is resolution within days, suggesting most infants do not require ongoing treatment with anti seizure medicines following discharge.
Prompt investigations to initiate specific treatments are also required though most NAIS cases will not require intervention beyond the above neuroprotective strategies. If infection is suspected, a full septic work-up and initiation of antimicrobial treatment is required until culture results are available. Bacterial meningitis may cause both perforating and large vessel arteritis with ongoing stroke recurrence risk, prompting some to consider a course of corticosteroids for which a favorable safety profile already exists (Hernandez 2011, Dunbar 2018, Siddiq 2017). A neonate with an abnormal cardiac examination should undergo echocardiography which may reveal structural anomalies suggesting a high recurrence risk, prompting consideration of anticoagulation.
Acute thrombophilia testing has a limited clinical value and may be difficult to interpret in the neonate (Lehman 2017, Curtis 2017, Monagle 2006). Acute thrombophilia testing might only be considered if there are other signs or symptoms of an underlying coagulation disorder. Hyperacute therapies, such as thrombolysis or mechanical thrombectomy, have no role in NAIS since the precise timing of the stroke is rarely known and endovascular devices are not designed for the small arteries of neonates (Ferrirero, 2019). Initiation of anticoagulation with low-molecular-weight heparin or unfractionated heparin, or starting antiplatelet therapy using aspirin, are rarely indicated in NAIS/PAIS because of the overall low risk of recurrence. The indication for initiation of anticoagulation or antiplatelet therapies would the presence of thrombophilia, or be the presence of congenital heart disease, with the exception of patent foramen ovale (PFO) (Ferreiro, 2019)
While there are no strong evidence-based protocols for the acute management of neonates with stroke, an example of a possible approach is shown in Figure 3.
NEONATAL CEREBRAL SINOVENOUS THROMBOSIS
Cerebral sinovenous thrombosis (CSVT) is the formation of a thrombus within the cerebral veins and/or dural sinuses within the first 28 days of life. Neonatal CSVT is less common than NAIS, occurring in approximately 1:9000 live births (Dunbar, 2020).
PATHOPHYSIOLOGY AND RISK FACTORS
Like most perinatal stroke diseases, the mechanisms of CSVT are often not clearly understood. Principles of venous thrombosis may include contributions of endothelial injury, hypercoagulable states, or venous stasis as dictated by Virchow’s triad (Ichord, 2017). Though not a stroke itself, CSVT may impair venous drainage from the brain, resulting first in venous edema but eventually venous infarction which often undergoes secondary hemorrhagic transformation. Where this occurs in the brain requires awareness of the venous anatomy including the superficial (superior sagittal sinus draining mostly cortex), deep (internal cerebral, medullary, and straight sinus draining most central brain structures), and common (transverse and sigmoid sinuses) systems.
With no controlled studies to date, risk factors for neonatal CSVT are not well defined. A majority of cases will be idiopathic though several likely primary etiologies can be recognized. Foremost among these is infection, including bacterial meningitis but also systemic sepsis. Dehydration may predispose newborns to CSVT, particularly those with a slow start to feeding who may develop a fluid deficit in the first week. Possible associations with systemic illness, cardiac disease or surgery, coagulopathy including thrombophilia, and mechanical compression are also considered. (deVeber 2001, Wu 2002, Fitzgerald 2006).
DIAGNOSIS
Clinical presentation of neonatal CSVT may be similar to other perinatal strokes but can also be more protean in nature. Seizures, both focal and generalized, and encephalopathy are common. Venous thrombosis is found more frequently in superficial system (deVeber 2001), possibly contributing to these cortical presentations. However, neonates may also present with less specific signs of encephalopathy including progressive coma, tonic posturing, and signs of increased intracranial pressure. Such signs are common in deep system CSVT where bilateral lesions to the thalami and hemorrhagic transformation with extension to large intraventricular hemorrhage and hydrocephalus (Kirton 2015). In fact, intraventricular hemorrhage in a term infant should be considered deep CSVT until proven otherwise (Kersbergen 2011, Wu 2003, Kirton 2015).
While large CSVT, infarcts, and hemorrhages as outlined above can be detected with cranial ultrasound and CT, MRI is again the investigation of choice for maximal sensitivity and specificity. Diffusion MRI can help distinguish vasogenic from cytotoxic edema (though the latter may be reversible in CSVT) and can demonstrate specific changes in discrete venous territories such as the medullary veins or the deep veins of the frontal white matter (so-called Iris sign). (Kersbergen, 2011). Hemorrhage-sensitive sequences can demonstrate venous congestion and early hemorrhagic transformation. MR venography (MRV) using neonatal modalities can be both sensitive and specific for the diagnosis of neonatal CSVT.
MANAGEMENT
With a high risk of progression, potentially severe morbidity, and safe treatments readily available, CSVT should be considered a directly treatable condition. There is significant variability in treatment of CSVT around the world though rates have been steadily increasing (Jordan, 2010). The largest prospective study of 160 neonates and children with CSVT demonstrated that non-treatment was associated with thrombus propagation in 28% of neonates (compared to <5% in those treated) with favorable safety of anticoagulation (Moharir 2010). Treatment with anticoagulation was not associated with increased risk of serious bleeding, while higher rates of infarction and poorer outcome were seen in the untreated group. The most recent scientific statement from the American Heart Association supports the consideration of anticoagulation with LMWH or unfractionated heparin for neonates with CSVT, particularly in the absence of hemorrhage, as well as in neonates with clinical deterioration or radiologic evidence of thrombus propagation. For those neonates who are not initially treated with anticoagulation, serial imaging should be performed at five to seven days to exclude thrombus propagation (Ferriero, 2019). In addition to consideration of anticoagulation, any treatable risk factors must be immediately addressed (antibiotics for infection, volume replacement for dehydration) and the principles of neuroprotection applied as outlined above.
NEONATAL HEMORRHAGIC STROKE
Neonatal hemorrhagic stroke (NHS) is a focal accumulation of blood within the brain parenchyma. By this definition, NHS includes primary hemorrhage as a distinct disease but also hemorrhagic transformation of ischemic injuries. The birth prevalence of NHS is approximately 1:6700 live births (Dunbar, 2020). Extra-axial hemorrhages including subarachnoid, subdural, and epidural are not considered within this definition of NHS. These hemorrhages are common and related to birth (Looney, 2007).
PATHOPHYSIOLOGY AND RISK FACTORS
Mechanisms of NHS involve the compromise of cerebrovascular integrity that results in blood entering the brain. Levels of evidence to understand causation are modest with most studies being single-center case series and only two population-based, controlled studies totaling about 100 cases combined (Armstrong-Wells 2009, Cole 2017). Consistent among these studies is that a specific cause can only be identified for a minority of cases. Such definitive causes include vascular malformations (arteriovenous malformations, aneurysms, cavernous malformations) and bleeding diatheses (e.g., neonatal alloimmune thrombocytopenia, hemophilia, vitamin K deficiency or refusal). This leaves the majority of cases as idiopathic NHS which are usually single, focal bleeds occurring near the time of birth and with what appears to be a very low recurrence rate. The only consistent possible risk factors across the controlled studies are clinical markers of fetal distress or difficult transition to life, which are non-specific. Birth “trauma” is often considered but in the only study to carefully measure this variable, no association was suggested (Cole, 2017). Common obstetrical factors such as induction, instrumentation, or caesarean section have not been established as independent risk factors for NHS (Towner 1999, Cole 2017) and must be considered accordingly.
DIAGNOSIS
NHS often presents with the same seizures or encephalopathy seen in other acute perinatal stroke diseases (Bruno 2014, Cole 2017). Bleeds that are large or in eloquent areas such as the posterior fossa may present with rapid neurological decline and signs of increased intracranial pressure. Such presentations require emergent imaging where cranial ultrasound or CT may be employed quickly and have high sensitivity for critical intracranial bleeds. For the stable neonate, MR is again the optimal imaging modality to confirm and characterize NHS (Figure 1C).
Most cases will demonstrate a single hematoma within the brain parenchyma where standard anatomical images (T1, T2) can help age the time of bleeding. Hemorrhage-sensitive sequences (GRE, SWI) not only confirm hemorrhage but have high sensitivity to detect small areas of additional bleeding which may inform potential etiologies. For example, identification of multifocal bleeds may suggest bleeding diathesis over a structural lesion. The temporal lobe is the most commonly affected location, but hemorrhages may occur anywhere (Cole, 2017). Vascular imaging with MRA and MRV may help define larger vascular malformations with implications for management and prognosis.
MANAGEMENT
Following critical stabilization of the newborn, urgent neurosurgical consultation may be indicated. Only a small minority of cases require immediate surgical intervention, and options may include decompression of large hematomas or insertion of extraventricular drains for obstructive or post-hemorrhagic hydrocephalus. Reversible causes must also be immediately excluded by performing CBC to confirm normal platelets and coagulation studies to exclude common inherited or acquired bleeding disorders. Supplemental Vitamin K should be considered if administration cannot be confirmed or was refused, or there is suspected deficiency secondary to in-utero exposures to maternal medications such as warfarin, phenytoin, or barbiturates (Ferriero, 2019). Head circumference should be followed closely. Fundamental principles of neuroprotection outlined above are essential in NHS. Advice of a hematologist is valuable to help exclude rarer forms of bleeding diathesis and gain advice on optimal replacement strategies for neonates with disordered coagulation. Genetic testing may be required for such bleeding disorders or vascular malformations (e.g., hereditary hemorrhagic telangiectasia). Follow-up imaging after 6 weeks once the hematoma has resolved is required to exclude smaller underlying vascular malformations.
ARTERIAL PRESUMED PERINATAL ISCHEMIC STROKE
Many perinatal stroke diseases will present outside the neonatal period. The most common presentation is an early hand preference or other asymmetry of motor development recognized by the parents at 4-6 months (Kirton, 2008). There is often a delay of 6-12 months after multiple referrals and, ultimately, an MRI that reveals the underlying stroke (Kirton, 2010). In many cases, the MRI will reveal a chronic arterial infarct with well circumscribed cystic encephalomalacia confirming to an arterial territory, most often the MCA (Figure 1D). Such arterial presumed perinatal ischemic stroke (APPIS) occurs in approximate 1:7800 live births (Dunbar, 2020). As such lesions are indistinguishable from chronic NAIS injuries, it is likely that these represent the same disease, differing only in the timing of presentation (i.e., seizures did not occur or were unrecognized in the neonatal period). The presentation as hemiparetic cerebral palsy dictates a selection bias towards APPIS lesions that affect the motor system, though other presentations may be recognized later in childhood such as visual field deficits or remote symptomatic epilepsy. In a similar fashion, presumed perinatal hemorrhagic stroke (PPHS) has also been described but appears to be very rare (Cole, 2017).
PERIVENTRICULAR VENOUS INFARCTION
The most common presumed perinatal stroke disease is periventricular venous infarction (PVI) with a birth prevalence of approximately 1:6000 live births (Dunbar, 2020). Long recognized as a common lesion in hemiparetic CP populations, the mechanism of PVI has been appreciated in recent years thanks to advanced imaging. PVI occurs in the fetus where germinal matrix hemorrhage, presumably prior to 32 weeks’ gestation. leads to secondary infarction of the periventricular white matter (Takanashi, 2005, Kirton 2008). The resulting lesion are clearly distinguishable from arterial strokes on MRI, with focal encephalomalacia limited to the periventricular white matter sparing the cortex and deep grey matter and, often, evidence of residual subependymal blood on susceptibility sequences (Kirton 2010) (Figure 1E). There have been no controlled studies of possible risk factors for PVI but recent unpublished data from the Canadian CP Registry suggests there is no association with perinatal factors, as would be expected given that the stroke is presumed to have occurred months before birth (Vitagliano, under review).
Due to symptomatic hemiparesis leading to selection for imaging, most children with PVI will have injuries to the corticospinal tracts. Although cortical structural changes may be seen above PVI lesions, the vast majority of children will have pure motor deficits and seizures are rare (Kirton, 2008). Mutations in COL4A1, the gene encoding a subunit of the type IV collagen, can create PVI-like lesions given its role in angiogenesis. Therefore, genetic testing should be considered when bilateral lesions or positive family history are present.
OUTCOME AND HABILITATION
Most survivors of perinatal stroke incur lifelong morbidity. Motor deficits, most commonly spastic hemiplegic cerebral palsy, predominate, but cognitive/behavioral disorders and epilepsy are also common (Kirton, 2013). Most outcome data come from studies of arterial ischemic stroke where approximately one third of children have normal outcome (deVeber 2000, Sreenan 2000, Chabrier 2016). The ability to predict outcome after perinatal stroke is dependent on multiple factors with only motor disability being highly predictive based on lesion characteristics. In survivors with abnormal outcome, multiple comorbidities are present in at least half (Sreenan 2000, deVeber 2003) although prevalence of severe disabilities is low (Chabrier 2016) and usually restricted to those with bilateral lesions (Mineyko, 2019).
Hemiparetic or unilateral cerebral palsy is the leading morbidity after perinatal stroke which is responsible for most cases (Lehman 2014). The risk of cerebral palsy is highly predictable from the diagnostic MRI, being more likely with larger strokes that include the motor cortex and/or corticospinal tract (Lehman 2014, Wu 2006, de Vries 2011). As shown by De Vries, specifically the involvement of the posterior limb of internal capsule on the term equivalent brain MRI has a highly positive predictive value for development of cerebral palsy (de Vries, 2011). However, the relative severity, distribution, and functional disability that results are variable across individuals. Much of this may relate to the developmental plasticity that occurs in the motor system following early unilateral injury which is increasingly well understood (Hilderley, 2019). Preservation of ipsilateral corticospinal connections from the non-lesioned hemisphere to the affected limbs creates a complex bi-hemispheric motor network that affects clinical motor function. Robotic studies have also characterized complex sensory deficits that further impair upper limb functions (Kuczynski, 2016). Remarkably, much more widespread alterations in the networks of the non-lesioned hemisphere of children with perinatal stroke are now being appreciated (Craig, 2020). Despite these advances in understanding post-stroke developmental plasticity, treatment options remain limited and imprecise. Constraint-induced movement therapy (CIMT) and bimanual therapies are effective for some children, but effect sizes are modest and response predictors remain undefined (Hoare, 2019). Hemiparetic children require early assessments and ongoing follow-up by occupational and physical therapists to consider options for support (orthoses, tone management), active lifestyle, and personalized, goal-directed therapy interventions to help individuals achieve maximum participation in life.
Despite what are often large lesions, only about 25% of children with unilateral stroke develop cognitive impairments (Kirton 2013, Murias, 2014). Language development is usually normal or near normal regardless of the side of the lesion (Kirton 2013; Ballantyne, 2008). Intellectual impairment and learning disabilities may not become apparent until school age, when impairments in complex neuropsychological functions become appreciable. Children with perinatal stroke require careful developmental surveillance through the preschool and early school age and should be referred for neuropsychological evaluations and supported education when concerns are recognized. Predictors for cognitive outcome are not well established but a recent retrospective cohort study found that recovery of the background aEEG pattern and cerebral oxygenation values measured by NIRS were associated with early neurocognitive development after correction for the size of the lesion (Wagenaar, 2019). The association of poor cognitive outcomes with epilepsy is also increasingly recognized (van Buuren 2013, Mineyko 2020).
Remote symptomatic epilepsy is a common consequence of perinatal stroke. Recent systematic reviews and population-based studies have agreed the occurrence is probably 25-30% (Billinghurst 2016, Rattani, 2019). Risk factors other than the presence of neonatal seizures are not well known (Fox 2016, Rattani 2019). Epilepsy can be refractory in approximately 25% of perinatal stroke survivors who are often good surgical candidates, especially if an invasive monitoring is used to direct focal cortical resections, lobar or multilobar resections/disconnections, or hemispherotomies (Ghatan, 2014). Epileptic encephalopathy must also be carefully considered, particularly sleep-related disorders such as continuous spike and wave in sleep (CSWS) (Mineyko, 2017). With such conditions having clear adverse effects on neurodevelopment but also being imminently treatable, careful surveillance is required and any child with perinatal stroke who has abnormal non-motor development, particularly in their preschool years, should have a sleep EEG to exclude this possibility. Early hemispherectomy for refractory seizures may be associated with improved cognitive and developmental outcomes (Wiebe 2013, Taussig, 2015).
Studies on the effects of perinatal stroke on vision are limited. Affected children will have significant variability in deficits based on location and extent of the injury, most often involving MCA syndromes, as well as posterior cerebral artery (PCA) strokes which occur in 8-15%. (Mercuri 2004, van der Aa 2013). A review study including 136 children with perinatal stroke found the most common visual abnormality was a visual field defect, however; incidence could not be determined as some studies did not include enough specific visual data (Crawford, 2018). Other visual deficits including “blindsight” (perception of visual stimuli without functional vision), visual cancellation errors, orthoptic eye movement abnormalities hemispace bias, and crowding acuity deficits have also been described (Crawford, 2018). There are few proven therapies for such conditions, but visual deficits should be considered for driving, school, and other adaptive supports for daily living.
Children with the other perinatal stroke diseases are at risk for similar complications to those described above for NAIS, with the following additional considerations. CVST may have very wide ranges of outcome, from no brain injury whatsoever to severe, bilateral deep injuries which can be devastating. The extent of infarction and presence of acute seizures have been associated with poor outcome from neonatal CSVT (deVeber, 2001). Outcomes from neonatal hemorrhagic stroke also vary widely with higher early mortality in addition to the possible sequelae described above, but recurrence is rare (Brouwer 2010, Armstrong-Wells 2009, Cole 2017). Perhaps related to the more random location of NHS lesions remote from the motor system, physical disabilities are less common as compared to ischemic perinatal stroke, present in 10-20%. Impairments in language, cognitive, and behavioural outcomes as well as epilepsy are avoided by the majority though long-term studies with school-aged assessments are lacking. For the presumed perinatal strokes, the vast majority have cerebral palsy (as this is usually the presenting sign) but while APPIS children likely have similar outcomes to NAIS, those with PVI usually avoid non-motor complications with favourable cognitive and developmental outcomes and low risk of epilepsy (Kirton, 2008).
PATIENT AND FAMILY MENTAL HEALTH AND SUPPORT
Parental morbidity is a substantial but under-recognized consequence of perinatal stroke. Perinatal stroke impacts the child, parents and caregivers across complex aspects of life and over the child’s lifespan. Many parents and their children demonstrate marked resiliency but are also at risk of long-term psychological morbidity. Increased rates of anxiety, depression and other mental health concerns including post-traumatic stress disorder have occurred among caregivers of children with perinatal stroke (Bemister 2014; Bemister 2015) and other forms of cerebral palsy (Pousada, 2013). Gender differences in how female and male caregivers perceive and cope with stress have also been observed (Matud, 2015). Mothers of children with moderate and severe disabilities following perinatal stroke tend to have an increased risk of depression, decreased marital satisfaction, poorer health-related quality of life and poorer family functioning, reflecting the need for additional resources and services (Bemister, 2015). Misplaced maternal guilt surrounding the causation of their child’s stroke may be a major contributor to these poor mental health outcomes. It is therefore incumbent upon treating health care providers to provide simple, factual information to mitigate this unfounded guilt, beginning at the time of diagnosis and revisited during follow-up. Free online videos are now available to facilitate this process (www.perinatalstroke.ca). Additional online resources (Family Guide to Pediatric Stroke, 2014) and support systems (https://iapediatricstroke.org/) are increasingly available for families through national and international pediatric stroke associations.
FUTURE DIRECTIONS
Despite marked progress in recent years, many challenges remain if outcomes from perinatal stroke are to be improved. Until the pathophysiology of perinatal stroke is better understood, we cannot develop preventative strategies. Controlled examination of placenta pathologies remains a major limitation that might be overcome by large-scale tissue banking studies. A recent study identified unique inflammatory cytokine profiles in newborns with NAIS (Mineyko, 2020), but whether this could be detected in thousands of expectant mothers to affect earlier delivery and stroke prevention seems unlikely. Therapeutic hypothermia has well-established benefits in neonatal hypoxic-ischemic encephalopathy and animal models of perinatal stroke but the inability to administer this therapy within hours of stroke occurrence will likely prevent any human clinical trials. More promising are animal studies and emerging neuroprotective trials of erythropoietin in humans with NAIS (Benders, 2014).
With limited options for prevention and protection, perinatal strokes will continue to occur, mandating a focus on neurorehabilitation to improve outcomes. As a focal injury in an otherwise healthy brain, perinatal stroke presents an ideal human model of developmental plasticity (Kirton, 2015). Human models may be defining personalized brain targets for non-invasive neuromodulation which has now entered phase 3 trials in children with perinatal stroke (Craig, 2020; Hilderley, 2019; Kirton 2016; Kirton, 2017). Novel forms of neurostimulation that might be simply applied in preschool age children in the home are also under investigation (Hollis A, 2020). In contrast to adults and older children, neonatal mechanisms of stroke recovery may invoke developmental neuroanatomical reorganization, the basic science of which may provide insights toward novel interventions (Kolb, 2011). The role of less studied and potentially modifiable co-morbidities merits particular attention including visuospatial neglect, visual impairments, fatigue, and epileptic encephalopathy. While early neuroimaging can predict motor outcome, the ability to predict complex cognitive outcomes remains limited, preventing the development of early intervention strategies. Preclinical research and early trials suggest safety and possible actions of growth factors and stem cells, though these face many challenges of cell-based therapies and require long-term outcomes to show efficacy (Wagenaar, 2017). Finally, engagement of families and those with lived experience to optimize supportive interventions and inform the research agenda should remain a top priority.
Review
ACUTE NEONATAL STROKE SAMPLE PROTOCOL
Arterial Ischemic
Stroke (NAIS)
Cerebral Sinovenous
Thrombosis (CSVT)
Hemorrhagic Stroke
(NHS)
Signs indicating potential stroke
- Seizures
- Altered Mental Status
- Abnormal Tone
- Focal Signs
- Apnea
- Acute neurological deterioration
Imaging: Neonatal MR Protocol (Sequences: DWI/ADC, GRE/SWI, T1, T2, MRA, MRV)
Consider TOF MRA Neck: to exclude carotid dissection
If MRI not available or contraindicated:
CT head +/- CTA or CTV
Neuroprotective Care
Maintain Normothermia, normovolemia, normoglycemia
Avoid direct pressure over occiput (especially in CSVT)
Consider continuous video EEG (or aEEG if conventional continuous EEG is not available)
Treat seizures, clinical and electrographic
NOTIFY NEUROCRITCAL CARE TEAM
| Arterial Ischemic Stroke (NAIS) | Cerebral Sinovenous Thrombosis (CSVT) | Hemorrhagic Stroke (NHS) |
Stroke-specific investigations | ||
Cardiac Echo Placental pathology | Coagulation work up (CBC, PTT/INR) | |
Stroke-specific Management | ||
Consider anticoagulation (ACT) if ongoing risk of cardiac embolism | Initiate anticoagulation unless contraindicated* | Neurosurgery consult
|
Additional Imaging | ||
| Usually not indicated Consider, if original study incomplete, or unexpected concerns | MR/MRV + GRE/SWI in 5-7 days if no ACT to exclude propagations. Otherwise in 6 weeks if ACT and stable | MR/MRV + GRE/SWI In 6-12 weeks or PRN |
Coagulation Testing will be determined by Hematology:
Newborn: Prot C, S and antithrombin, PT/PTT
(Factor V Leiden and Prothrombin G20210A at 3 mo of age)
Maternal testing: ANA, Lupus, Anticardiolipin Ab, Homocysteine
Both parents: Prot S and C
Family Education
Review evidence-based outcome
Provide Parent Guide to Pediatric Stroke, web-based resources (IPSO, IAPS)
Follow up
Neurocritical Care phone follow-up 2 weeks after discharge
Refer to perinatal stroke clinic (4-6 weeks for CSVT)
Provide contact information (stroke neurologist, Neurocritical NP phone #, pediatric neurology nursing phone #)
Determine the need for coagulation testing
Any of the following present:
Confirmed CVST
OR
Strong family hx of thrombotic disease
OR
Multiple thrombosis sites
OR
Large burden of thrombosis
Literature
- Raju TNK, Nelson KB, Ferriero D, Lynch JK, NICHD-NINDS Perinatal Stroke Workshop Participants. Pediatrics. 2007 Sep;120(3):609-16. Doi:10.1542/peds.2007-0336
- Martinez-Biarge M, Ferriero DM, Cowan FM: Perinatal arterial ischemic stroke. Chapter 11. Handbook of Clinical Neurology. Vol 162 (3rd series) 2019
- Dunbar M, Kirton A. Perinatal Stroke: mechanisms, management, and outcomes of early cerebrovascular brain injury. Lancet Child Adolesc Health. 2018 Sep;2(9):666-676. doi:10.1016/S2352-4642(18)30173-1.
- Dunbar M, Mineyko A, Hill M, Hodge J, Floer A, Kirton A. Population Based Birth Prevalence of Disease-Specific Perinatal Stroke. Pediatrics. 2020 Nov;146(5):e2020013201. doi: 10.1542/peds.2020-013201
- Armstrong-Wells J, Johnston SC, Wu YW, Sidney S, Fullerton HJ. Prevalence and predictors of perinatal hemorrhagic stroke: results from the kaiser pediatric stroke study. Pediatrics. 2009;123(3):823.
- Prenatal Stroke. Semin Fetal Neonatal Med. 2009 Oct;14(5):250-66. doi: 10.1016/j.siny.2009.07.008
- Golomb MR, Dick PT, MacGregor DL, Armstrong DC, deVeber GA: Cranial ultrasonography has a low sensitivity for detecting arterial ischemic stroke in term neonates. J Child Neurol 2003 Feb;18(2): 98-103
- Lehman L, Beaute BA, Kapur K, Danehy AR, Benson-Leung ME, Malkin BA, Rivkin MJ, Trenor CC III. Workup for perinatal stroke does not predict recurrence. Stroke. 2017; 48: 2078-2083. Doi:10.1016/STROKEAHA.117.017356
- Ferriero D, Fullerton HJ, Bernard TJ, Billinghurst L, Daniels SR, DeBaun MR, deVeber G, Ichord RN, Jordan LC, Massicotte P, Meldau J, Roach ES, Smith ER on behalf of the American Heart Association Stroke Council and Council on Cardiovascular and Stroke Nursing. Stroke. 2019 50:e51-e96. DOI:10.1161/STR0000000000000183
- Dunbar M, Kirton A. Perinatal Stroke. Seminars in Pediatric Neurology. 2019;32:100767. doi:10.1016/j.spen.2019.08.003
- Lee J, Croen LA, Backstrand KH, Yoshida CK, Henning LH, Lindan C, Ferriero DM, Fullerton HJ, Barkovich AJ, Wu YW. Maternal and infant characteristics associated with perinatal arterial stroke in the infant. JAMA 2005 Feb 9;293(6):723-9. doi:10.1001/jama.293.6.723
- Elbers J, Viero S, MacGregor D, DeVeber G, Moore AM. Placental pathology in neonatal stroke. Pedatrics. 2011:127:e722-e729. Doi:10.1542/peds.2010-1490
- Sorg Anna-Lisa, von Kries R, Klemme M, Gerstl L, Weinberger R, Beyerlein A, Lack N, Felderhoff-Musser U, Dzietko M. Risk factors for perinatal arterial ischemic stroke: a large case-control study. Developmental Medicine and Child Neurology 2020, 62: 513-520. DOI: 10.1111/dmcn.14347
- Roy B, Arbuckle S, Walker K, Morgan C, Galea C, Badawi N, Novak I. The role of the placenta in perinatal stroke: A systematic review. J Child Neurol. 2020 Oct;35(11):773-783. doi:10.1177/0883073820929214
- Mineyko A, Kirton A. The black box of perinatal ischemic stroke pathogenesis. J Child Neurol. 2011;26(9):1154-1162. doi:https://doi.org/10.1177/0883073811408312
- Nelson KB, Lynch JK. Stroke in newborn infants. Lancet Neurol. 2004; 3:150-158. Doi: 10.1016/S1474-4422(04)00679-9.
- Wu YW, Lynch JK, Nelson KB. Perinatal arterial stroke: understanding mechanisms and outcomes. Semin Neurol. 2005 Dec;25(4):424-34. doi:10.1055/s-2005-923536.
- Curtis C, Mineyko A, Massicotte P, Leaker M, Jiang XY, Floer A, Kirton A. Thrombophilia risk is not increased in children after perinatal stroke. Blood. 2017 May 18;129(20):2793-2800. doi: 10.1182/blood-2016-11-750893
- Curtis, Colleen, Aleksandra Mineyko, Patricia Massicotte, Michael Leaker, Xiu Yan Jiang, Amalia Floer, and Adam Kirton. “Thrombophilia Risk Is Not Increased in Children after Perinatal Stroke.” Blood, March 3, 2017. https://doi.org/10.1182/blood-2016-11-750893.
- Kirton A, deVeber G. Pediatric stroke: pressing issues and promising directions. Lancet Neurol. 2015; 14: 92-102
- Akanli LF, Trasi SS, Thuraisamy K, Bergtraum MP, Thantu A, Fischer RF, Cohen-Addad N. Am J Perinatol. 1998 Jun;15(6):399-402. doi: 10.1055/s-2007-993965.
- Martinez-Biarge M, Cheong JLY, Diez-Sebastian J, Mercuri E, Dubowitz LMS, Cowan FM. Risk factors for neonatal arterial ischemic stroke: the importance of the intrapartum period. J Pediatr. 2016 Jun:173:62-68.e1. doi:10.1016/j.peds.2016.02.064
- Darmency-Stamboul V, Chantegret C, Ferdynus C, Mejean N, Durand C, Sagot P, Giroud M, Bejot Y, Gouyon JB. Stroke, 2012 Sep;43(9):2307-12. doi:10.1161/STROKEAHA.111.642181
- Chasnoff IJ, Bussey ME, Savich R, Stack CM. Perinatal cerebral infarction and maternal cocaine use. J Pediatr. 1986 Mar;108(3):456-9. doi:10.1016/s0022-3476(86)80896-4
- Cheong JLY, Cowan FM. Neonatal arterial ischemic stroke: obstetric issues. Semin Fetal Neonatal Med. 2009 Oct;14(5):267-71
- Günther G, Junker R, Sträter R, Schobess R, Kurnik K, Kosch A, Nowak-Göttl U, Childhood Stroke Study Group. Symptomatic ischemic stroke in full-term neonates: role of acquired and genetic prothrombotic risk factors. Stroke. 2000 Oct;31(10):2437-41. doi: 10.1161/01.str.31.10.2437
- Lequin MH, Peeters EA, Holscher HC, de Krijger R, Govaert P. Arterial infarction caused by carotid artery dissection in the neonate. Eur J Paediatr Neurol. 2004;8:155-160. Doi:10.1016/j.ejpn.2004.02.001.
- Elbers J, Viero S, MacGregor D, deVeber G, Moore AM. Placental pathology in neonatal stroke. Pediatrics. 2011;127:e722-e729.
- Harteman JC, Groenendaal F, Kwee A, Welsing PMJ, Benders MJNL, de Vries LS. Risk factors for perinatal arterial ischemic stroke in full-term infants: a case-control study. Arch Dis Child Fetal Neonatal Ed. 2012 Nov;97(6):F411-6. doi: 10.1136/archdischild-2011-300973.
- Mineyko A, Nettel-Aguirre A, de Jesus P, et al. Association of neonatal inflammatory markers and perinatal stroke subtypes. Neurology. Published online July 29, 2020. doi:10.1212/WNL.0000000000010309
- Takenouchi T, Ohyagi M, Torii C, Kosaki R, Takahashi T, Kosaki K. Porencephaly in a fetus and HANAC in her father: variable expression of COL4A1 mutation. Am J Med Genet A. 2015 Jan;167A(1):156-8.
- Rafay MF, Pontigon AM, Chiang J, et al. Delay to diagnosis in acute pediatric arterial ischemic stroke. Stroke. 2009;40:58-64.
- Lee S, Mirsky DM, Beslow LA, et al. Pathways for Neuroimaging of Neonatal Stroke. Pediatr Neurol. 2017;69:37-48. doi:10.1016/j.pediatrneurol.2016.12.008
- Sharpe C, Reiner GE, Davis SL, et al. Levetiracetam Versus Phenobarbital for Neonatal Seizures: A Randomized Controlled Trial. Pediatrics. 2020;145(6):e20193182. doi:10.1542/peds.2019-3182
- Hernandez MI, Sandoval CC, Tapia JL, et al. Stroke patterns in neonatal group B streptococcal meningitis. PediatrNeurol. 2011;44:282-288.
- Dunbar M, Shah H, Shinde S, et al. Stroke in Pediatric Bacterial Meningitis: Population-Based Epidemiology. Pediatr Neurol. 2018;89:11-18. doi:10.1016/j.pediatrneurol.2018.09.005
- Siddiq Ishita, Armstrong Derek, Surmava Ann-Marie, Dlamini N, MacGregor D, Moharir M, Askalan R. Utility od Neurovascular Imaging in Acute Neonatal Arterial Stroke. J Pediatr 2017; 188:110-4
- Monagle P, Barnes C, Ignjatovic V, et al. Developmental haemostasis. Impact for clinical haemostasis laboratories. Thromb Haemost. 2006;95:362-372.
- Ichord R. Cerebral Sinovenous Thrombosis. Front Pediatr. 2017;5:163. doi:10.3389/fped.2017.00163
- deVeber G, Andrew M, Adams C, et al. Cerebral sinovenous thrombosis in children. N Engl J Med. 2001;345(6):417-423. doi:10.1056/NEJM200108093450604
- Wu YW, Miller SP, Chin K, et al. Multiple risk factors in neonatal sinovenous thrombosis. Neurology. 2002;59(0028-3878):438-440.
- Fitzgerald KC, Williams LS, Garg BP, Carvalho KS, Golomb MR. Cerebral sinovenous thrombosis in the neonate. ArchNeurol. 2006;63:405-409.
- Kersbergen KJ, Groenendaal F, Benders MJNL, et al. The spectrum of associated brain lesions in cerebral sinovenous thrombosis: relation to gestational age and outcome. Arch Dis Child Fetal Neonatal Ed. 2011;96(6):F404-409. doi:10.1136/adc.2010.201129
- Wu YW, Hamrick SE, Miller SP, et al. Intraventricular hemorrhage in term neonates caused by sinovenous thrombosis. Ann Neurol. 2003;54(0364-5134):123-126.
- Jordan LC, Rafay M, Smith S, Askalan R,Zamel K, deVeber G, Ashwal S. Antithrombotic treatment in neonatal cerebral sinovenous thrombosis. Results of the International Pediatric Stroke Study. J Pediatr 2010; 156:704-10.
- Kharoshankaya L, Stevenson N, Livingstone V, Murray D, Murphy B, Ahearne CE, Boylan G. Seizure burden and developmental outcome in neonates with hypoxic-ischemic encephalopathy. Dev Med Child Neurol. 2016 Dec;58(12):1242-1248. PMID 27595841. DOI 10.1111/dmcn.13215
- Shellhaas RA, Chang T, Tsuchida T, Scher MS, Riviello J, Abend NS, Nguyen S, Wusthoff CJ, Clancy RR. The American Clinical Neurophysiology Society’s Guideline on continuous electroencephalography monitoring in neonates. J Clin Neurophysiol. 2011 Dec28(6):611-7. PMID 22146359. DOI 10.1097/WNP.0b013e3182e96d7
- Moharir MD, Shroff M, Stephens D, et al. Anticoagulants in pediatric cerebral sinovenous thrombosis: a safety and outcome study. Ann Neurol. 2010;67:590-599.
- Looney CB, Smith JK, Merck LH, Wolfe HM, Chescheir NC, Hamer RM, Gilmore JH: Intracranial hemorrhage in asymptomatic neonates: prevalence on MR images and relationship to obstetricc and neonatal risk factors. Radiology. 2007 Feb; 242(2):535-41
- Armstrong-Wells J, Johnston SC, Wu YW, Sidney S, Fullerton HJ. Prevalence and predictors of perinatal hemorrhagic stroke: results from the kaiser pediatric stroke study. Pediatrics. 2009;123:823-828.
- Cole L, Dewey D, Letourneau N, et al. Clinical characteristics, risk factors, and outcomes associated with neonatal hemorrhagic stroke: A population-based case-control study. JAMA Pediatr. Published online January 17, 2017.doi:10.1001/jamapediatrics.2016.4151
- Towner D, Castro MA, Eby-Wilkens E, Gilbert WM. Effect of mode of delivery in nulliparous women on neonatal intracranial injury. N Engl J Med. 1999;341(23):1709-1714. doi:10.1056/NEJM199912023412301
- Bruno CJ, Beslow LA, Witmer CM, et al. Haemorrhagic stroke in term and late preterm neonates. ArchDisChild Fetal Neonatal Ed. 2014;99:F48-F53.
- Kirton A, deVeber G, Pontigon AM, MacGregor D, Shroff M. Presumed perinatal ischemic stroke: vascular classification predicts outcomes. Ann Neurol. 2008;63(4):436-443. doi:https://doi.org/10.1002/ana.21334
- Kirton A, Shroff M, Pontigon AM, deVeber G. Risk factors and presentations of periventricular venous infarction vs arterial presumed perinatal ischemic stroke. ArchNeurol. 2010;67:842-848.
- Takanashi J, Tada H, Barkovich AJ, Kohno Y. Magnetic resonance imaging confirms periventricular venous infarction in a term-born child with congenital hemiplegia. Dev Med Child Neurol. 2005;47:706-708.
- Kirton A, Wei X. Teaching neuroimages: confirmation of prenatal periventricular venous infarction with susceptibility-weighted MRI. Neurology. 2010;74(12):e48.
- Li D, Hodge J, Wei X-C, Kirton A. Reduced ipsilesional cortical volumes in fetal periventricular venous infarction. Stroke. 2012;43(5):1404-1407. doi:10.1161/STROKEAHA.111.645077
- Kirton A. Life After Perinatal Stroke. Stroke. 2013;44(11):3265-3271. doi:https://doi.org/10.1161/STROKEAHA.113.000739
- deVeber G, Canadian Paediatric Ischemic Stroke Study Group. Canadian Paediatric Ischemic Stroke Registry: Analysis of children with arterial ischemic stroke. Ann Neurol. 2000;48(3):514.
- Sreenan C, Bhargava R, Robertson CM. Cerebral infarction in the term newborn: clinical presentation and long- term outcome. JPediatr. 2000;137:351-355.
- Chabrier S, Peyric E, Drutel L, et al. Multimodal Outcome at 7 Years of Age after Neonatal Arterial Ischemic Stroke. J Pediatr. 2016;172:156-161.e3. doi:10.1016/j.jpeds.2016.01.069
- DeVeber G, MacGregor D, Friefeld S, Chan A, Meanery B, Domi T. Neurological Outcome in Survivors of Neonatal Arterial Ischemic Stroke. Pediatr Res. 2003;53(4):537A.
- Mineyko A, Kirton A. Long-Term Outcome After Bilateral Perinatal Arterial Ischemic Stroke. Pediatr Neurol. Published online August 2, 2019. doi:10.1016/j.pediatrneurol.2019.07.013
- Lehman LL, Rivkin MJ. Perinatal Arterial Ischemic Stroke: Presentation, Risk Factors, Evaluation, and Outcome. Pediatr Neurol. 2014;51(6):760-768. doi:10.1016/j.pediatrneurol.2014.07.031
- Wu YW, Lindan CE, Henning LH, et al. Neuroimaging abnormalities in infants with congenital hemiparesis. Pediatr Neurol. 2006;35(3):191-196
- Hilderley AJ, Metzler MJ, Kirton A. Noninvasive Neuromodulation to Promote Motor Skill Gains After Perinatal Stroke. Stroke. Published online January 21, 2019:STROKEAHA118020477. doi:10.1161/STROKEAHA.118.020477
- Kuczynski AM, Dukelow SP, Semrau JA, Kirton A. Robotic Quantification of Position Sense in Children With Perinatal Stroke. Neurorehabil Neural Repair. 2016;30(8):762-772. doi:10.1177/1545968315624781
- Craig BT, Hilderley AJ, Kinney-Lang E, Carlson HL, Kirton A. Developmental neuroplasticity of the white-matter connectome in children with perinatal stroke. Neurology. 2020;95(18):e2476-e2486. doi:10.1212/WNL.0000000000010669
- Hoare BJ, Wallen MA, Thorley MN, Jackman ML, Carey LM, Imms C. Constraint-induced movement therapy in children with unilateral cerebral palsy. Cochrane Database Syst Rev. 2019;4:CD004149. doi:10.1002/14651858.CD004149.pub3
- Murias K, Brooks B, Kirton A, Iaria G. A review of cognitive outcomes in children following perinatal stroke. Dev Neuropsychol. 2014;39(2):131-157. doi:10.1080/87565641.2013.870178
- Ballantyne AO, Spilkin AM, Hesselink J, Trauner DA. Plasticity in the developing brain: intellectual, language and academic functions in children with ischaemic perinatal stroke. Brain. 2008;131:2975-2985.
- Wagenaar N, van den Berk DJM, Lemmers PMA, et al. Brain Activity and Cerebral Oxygenation After Perinatal Arterial Ischemic Stroke Are Associated With Neurodevelopment. Stroke. Published online August 8, 2019:STROKEAHA119025346. doi:10.1161/STROKEAHA.119.025346
- van Buuren LM, van der Aa NE, Dekker HC, Vermeulen RJ, van Nieuwenhuizen O, van Schooneveld MM, de Vries LS. Cognitive outcome in childhood after unilateral perinatal brain injury. Dev Med Child Neurol. 2013 Oct;55(10):934-40. Epub 2013 Jun 13
- Mineyko A, Kirton A, Billinghurst L, et al. Seizures and Outcome One Year After Neonatal and Childhood Cerebral Sinovenous Thrombosis. Pediatr Neurol. 2020;105:21-26. doi:10.1016/j.pediatrneurol.2019.08.012
- Rattani A, Lim J, Mistry AM, et al. Incidence of Epilepsy and Associated Risk Factors in Perinatal Ischemic Stroke Survivors. Pediatr Neurol. 2019;90:44-55. doi:10.1016/j.pediatrneurol.2018.08.025
- Billinghurst LL, Beslow LA, Abend NS, et al. Incidence and predictors of epilepsy after pediatric arterial ischemic stroke. Neurology. 2017;88(7):630-637. doi:10.1212/WNL.0000000000003603
- Fox CK, Glass HC, Sidney S, Smith SE, Fullerton HJ. Neonatal seizures triple the risk of a remote seizure after perinatal ischemic stroke. Neurology. 2016;86(23):2179-2186. doi:10.1212/WNL.0000000000002739
- Ghatan S, McGoldrick P, Palmese C, Le Vega-Talbott M, Kang H, Kokoszka AM, Goodman RR, Wolf SM. Surgical management of medically refractory epilepsy due to early childhood stroke. J Neurosurg Pediatr. 2014 Jul;14(1):58-67. Doi:10.3171/2014.3.PEDS13440
- Mineyko A, Qi W, Carlson HL, Bello-Espinosa L, Brooks BL, Kirton A. Neuropsychological outcome in perinatal stroke associated with epileptiform discharges in sleep. Can J Neurol Sci J Can Sci Neurol. 2017;44(4):358-365. doi:10.1017/cjn.2017.29
- Wiebe S, Berg AT. Big epilepsy surgery for little people: what’s the full story on hemispherectomy? Neurology. 2013 Jan 15;80(3):232-3.
- Taussig D, Dorfmuller G, Save J, Fohlen M, Chipaux M, Ferrand-Sorbets S, Delalande O, Bulteau C. Hemispherectomy for isolated infantile spasms following perinatal ischemic stroke.
- Crawford LB, Golomb M. Childhood stroke and vision: A review of the literature. Pediatr Neurol. 2018. Apr;81:6-13. doi: 10.1016/j.pediatrneurol.2017.11.007.
- Mercuri E, Anker S, Guzzetta A, et al. Neonatal cerebral infarction and visual function at school age. ArchDisChild Fetal Neonatal Ed. 2003;88(1359-2998):F487-F491.
- van der Aa NE, Dudink J, Benders MJ, et al. Neonatal posterior cerebral artery stroke: clinical presentation, MRI findings, and outcome. Dev Med Child Neurol. 2013;55:283-29
- Brouwer MJ, de Vries LS, Pistorius L, Rademaker KJ, Groenendaal F, Benders MJNL. Ultrasound measurements of the lateral ventricles in neonates: why, how and when? A systematic review. Acta Paediatr Oslo Nor 1992. 2010;99(9):1298-1306. doi:10.1111/j.1651-2227.2010.01830.x
- Bemister TB, Brooks BL, Dyck RH, Kirton A. Parent and family impact of raising a child with perinatal stroke. BMC Pediatr. 2014;14:182. doi:10.1186/1471-2431-14-182
- Bemister TB, Brooks BL, Dyck RH, Kirton A. Predictors of caregiver depression and family functioning after perinatal stroke. BMC Pediatr. 2015;15:75. doi:10.1186/s12887-015-0397-5
- Pousada M, Guillamon N, Hernandez-Encuentra E, Munoz E, Redolar D, Boixados M, Gomez-Zuniga Impact of caring for a child with cerebral palsy on the quality of life of parents: a systematic review of the literature. J Dev Phys Disabil 2013, 25(5):545-577
- Benders MJ, van der Aa NE, Roks M, et al. Feasibility and safety of erythropoietin for neuroprotection after perinatal arterial ischemic stroke. J Pediatr. 2014;164(3):481-486.e1-2. doi:10.1016/j.jpeds.2013.10.084
- Craig BT, Hilderley A, Kirton A, Carlson HL. Imaging Developmental and Interventional Plasticity Following Perinatal Stroke. Can J Neurol Sci J Can Sci Neurol. Published online July 30, 2020:1-41. doi:10.1017/cjn.2020.166
- Kirton A, Andersen J, Herrero M, et al. Brain stimulation and constraint for perinatal stroke hemiparesis: The PLASTIC CHAMPS Trial. Neurology. 2016;86(18):1659-1667. doi:10.1212/WNL.0000000000002646
- Kirton A, Ciechanski P, Zewdie E, et al. Transcranial direct current stimulation for children with perinatal stroke and hemiparesis. Neurology. 2017;88(3):259-267. doi:10.1212/WNL.0000000000003518
- Hollis A, Zewdie E, Nettel-Aguirre A, et al. Transcranial Static Magnetic Field Stimulation of the Motor Cortex in Children. Front Neurosci. 2020;14:464. doi:10.3389/fnins.2020.00464
- Kolb B, Mychasiuk R, Williams P, Gibb R. Brain plasticity and recovery from early cortical injury. Dev Med Child Neurol. 2011;53 Suppl 4:4-8.
Wagenaar N, Nijboer CH, van Bel F. Repair of neonatal brain injury: bringing stem cell-based therapy into clinical practice. Dev Med Child Neurol. 2017;59(10):997-1003. doi:10.1111/dmcn.13528
Mailo Neonatal Stroke Review
